



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2022-10-21
Posting Date:2022-10-21 Views:
Views: Recently, Simcere Pharmaceutical secured the first generic of ibrutinib, whose original product achieved global sales of US$9.78 billion in 2021. Leveraging market and pricing advantages, first-generation generics have consistently been a key focus in the R&D strategies of domestic pharmaceutical companies. According to不完全统计, among the blockbuster drugs with global sales exceeding US$1 billion in 2021 that have not yet had a first generic approved in China, 19 varieties have generic drugs under development by pharmaceutical companies. Among these, 5 varieties had sales exceeding RMB 1 billion in 2021 across the three major terminals and six major markets (detailed scope at the end of this article).
From the perspective of corporate filings, China Biopharmaceutical has laid out plans for 8 varieties, among which ruxolitinib, everolimus, and fluticasone vilanterol are exclusive filings. Qilu Pharmaceutical has laid out plans for 7 varieties, with dolutegravir, riociguat, and olaparib being exclusive/first filings. Additionally, Kelun Pharmaceutical, Beite Pharmaceutical, and Xuantai Pharmaceutical have each laid out plans for 3 varieties.
Varieties with global sales exceeding US$1 billion in 2021 and no first generic approved in China yet
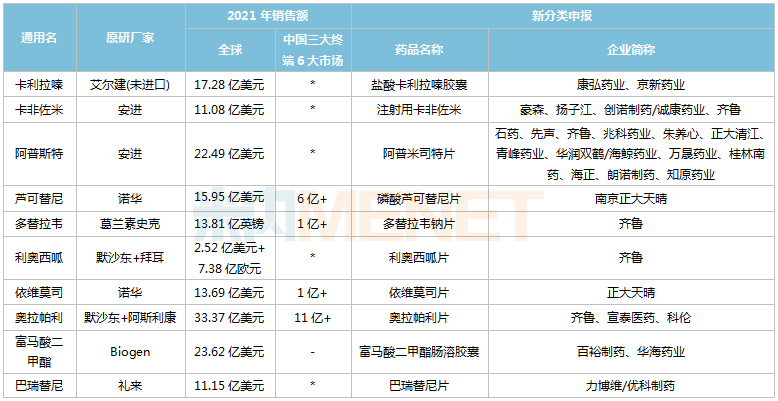
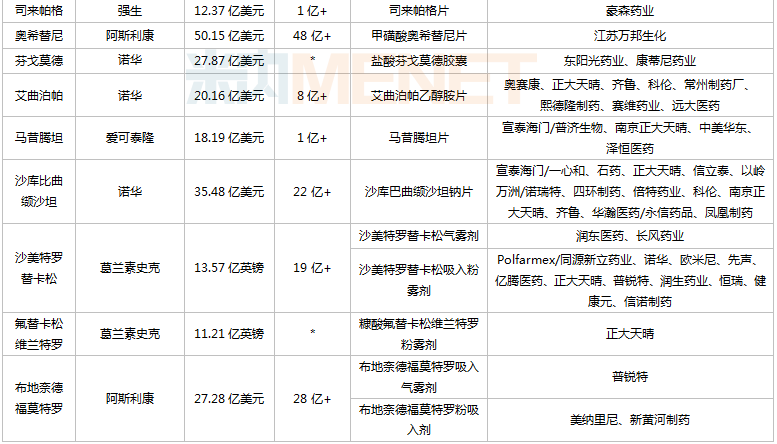
Note: No sales or sales below RMB 100 million are indicated with *
Osimertinib
Osimertinib is a third-generation, oral, irreversible selective EGFR mutation inhibitor developed by AstraZeneca. It was first approved by the FDA in November 2015, achieving global sales of US$5.015 billion in 2021, an increase of about 16% year-on-year.
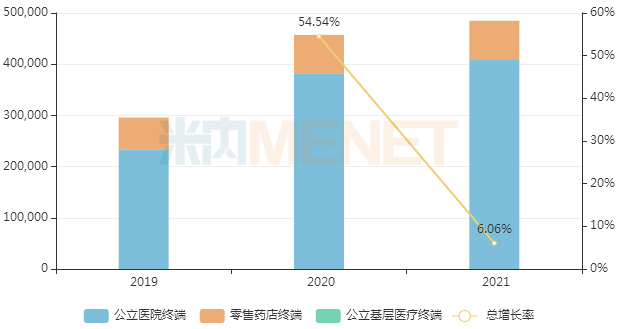
In March 2017, AstraZeneca's osimertinib was first approved for entry into the domestic market. It was included in the national medical insurance Category B list through negotiations in 2018, and the agreement was successfully renewed in 2020, with both first-line and second-line indications included, which is expected to further increase sales volume. According to Menet data, sales of osimertinib in China's three major terminals and six major markets in 2021 exceeded RMB 4.8 billion, a year-on-year increase of 6.06%.
Sales of osimertinib in China's three major terminals and six major markets in recent years (Unit: RMB 10,000)
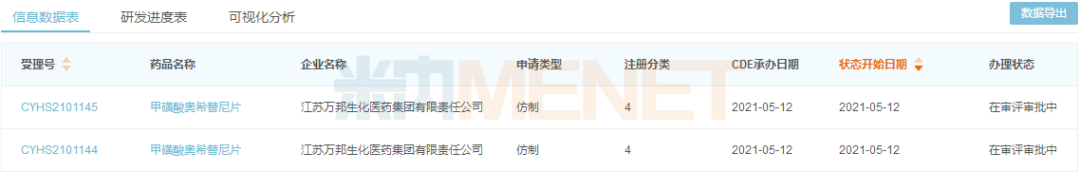
The domestic compound patent for osimertinib (CN103702990) expires in July 2032. Currently, only Jiangsu Wanbang Biochemical has filed for a generic version; its product completed a BE trial in August 2020 and submitted a marketing application under Category 4 of generic drugs in May 2021.
New classification filing status for Osimertinib Mesylate Tablets
Sacubitril Valsartan
Sacubitril valsartan is a combination preparation composed of sacubitril (a neprilysin inhibitor) and valsartan (an angiotensin receptor antagonist). It was first approved by the FDA in July 2015 and is the world's first breakthrough innovative drug in the field of heart failure treatment. Its global sales in 2021 reached US$3.548 billion, a year-on-year increase of about 42%.
In July 2017, Novartis's sacubitril valsartan was first approved for entry into the domestic market. It was included in the national medical insurance Category B list through negotiations in 2019, and the agreement was successfully renewed in 2021, with the addition of a new indication for primary hypertension. Menet data shows that in recent years, the sales growth rate of sacubitril valsartan in China's three major terminals and six major markets has been in the triple digits, exceeding RMB 2 billion for the first time in 2021, a year-on-year increase of 127.62%.
Sales of sacubitril valsartan in the three major terminals and six major markets in recent years (Unit: RMB 10,000)
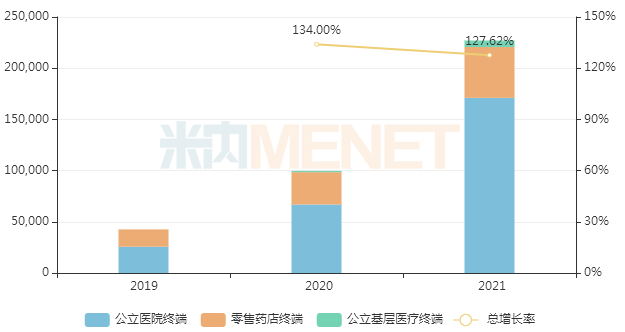
Although the patent for sacubitril valsartan has not yet expired, more than 10 domestic pharmaceutical companies have submitted marketing applications for this product under Category 4 of generic drugs. Among them, Shanghai Xuantai Haimen Pharmaceutical was the first to file, with its application accepted by the CDE in November 2018, and it is currently in the second round of data supplementation. Additionally, products from CSPC Ouyi, Chia Tai Tianqing, and other companies are in the first round of data supplementation.
New classification filing status for Sacubitril Valsartan Sodium Tablets

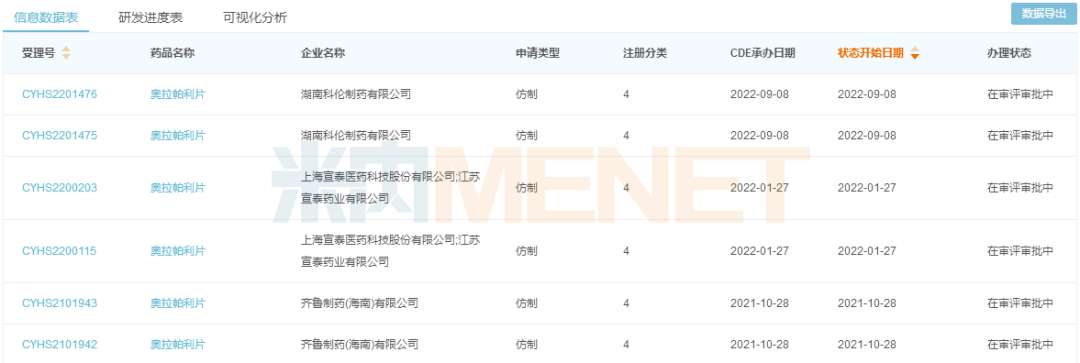
Olaparib
Olaparib belongs to the class of PARP inhibitors. It works by inhibiting PARP, thereby reducing or even preventing cancer cells with damaged BRCA genes from repairing DNA, ultimately leading to cancer cell death. Co-developed by AstraZeneca and Merck & Co., it was first approved by the FDA in December 2014 as the world's first PARP inhibitor, achieving global sales of US$3.337 billion in 2021.
In August 2018, AstraZeneca's olaparib was first approved for entry into the domestic market. It was included in the national medical insurance Category B list through negotiations in 2019, and the agreement was successfully renewed with additional indications in 2020. Menet data shows that in recent years, the sales scale of olaparib in China's three major terminals and six major markets has increased year by year, exceeding RMB 1 billion for the first time in 2021, a year-on-year increase of 23.08%.
Sales of olaparib in China's three major terminals and six major markets in recent years (Unit: RMB 10,000)
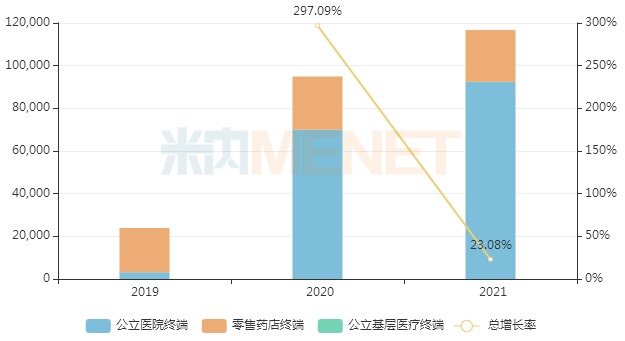
It is reported that the compound patent for olaparib will expire in 2024. Currently, three domestic pharmaceutical companies have submitted marketing applications for this product under the new registration classification, including Qilu Pharmaceutical, Xuantai Pharmaceutical, and Hunan Kelun Pharmaceutical. Among them, Qilu Pharmaceutical was the first to file and is currently in the first round of data supplementation.
New classification filing status for Olaparib Tablets
Budesonide Formoterol
Budesonide formoterol is a combination inhalation agent composed of budesonide (a corticosteroid) and formoterol (a long-acting β2-agonist), used to treat asthma, chronic obstructive pulmonary disease, and other conditions. Although the patent for budesonide formoterol has expired, its global sales did not experience a sharp decline, still reaching US$2.728 billion in 2021.
AstraZeneca's budesonide formoterol entered the domestic market relatively early. In recent years, its sales in China's three major terminals and six major markets have increased year by year, surpassing RMB 2 billion for the first time in 2018 and exceeding RMB 2.8 billion in 2021, a year-on-year increase of 10.35%.
Sales of budesonide formoterol in China's three major terminals and six major markets in recent years (Unit: RMB 10,000)
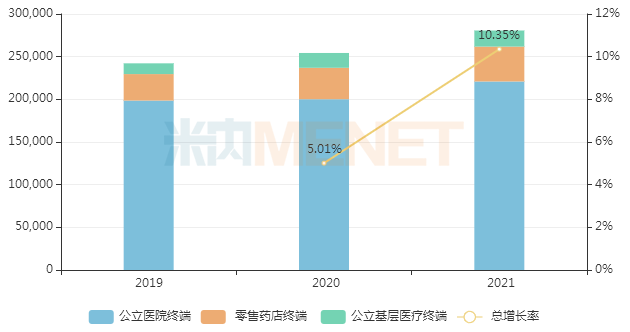
There are currently few companies developing generic versions of budesonide formoterol. Puruite (a subsidiary of Beite Pharmaceutical), which considers inhalation drugs a key track in China, was the earliest to develop a generic version. The company submitted a marketing application for Budesonide Formoterol Inhalation Aerosol under Category 3 of generic drugs in December 2017 and is currently conducting Phase III clinical trials.
New classification filing status for Budesonide Formoterol Inhalation

Salmeterol Fluticasone
Salmeterol fluticasone is a combination inhalation agent composed of salmeterol (a long-acting β2-agonist) and fluticasone (a corticosteroid), used to treat asthma, chronic obstructive pulmonary disease, etc. As one of GlaxoSmithKline's blockbuster products, global sales of salmeterol fluticasone exceeded US$5 billion in 2013. Although sales declined after the patent expired, it still exceeded US$1 billion in 2021.
GlaxoSmithKline's salmeterol fluticasone also entered the domestic market relatively early. In recent years, its sales in China's three major terminals and six major markets have fluctuated, approaching RMB 2 billion in 2021, a year-on-year increase of 6.23%.
Sales of salmeterol fluticasone in China's three major terminals and six major markets in recent years (Unit: RMB 10,000)
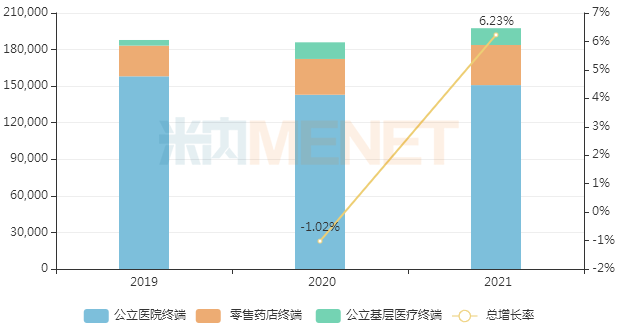
Currently, more than 10 pharmaceutical companies are developing generic versions of salmeterol fluticasone, including well-known companies such as Chia Tai Tianqing, Hengrui Medicine, and Health Element. However, due to high barriers to generic development, no generic version has been approved in China to date.
New classification filing status for Salmeterol Fluticasone Inhalation

Conclusion
Driven by policies such as the continuous advancement of consistency evaluation, the normalization of centralized procurement, and the regular adjustment of medical insurance, coupled with the comprehensive promotion of drug review reform, the introduction of the "patent linkage system" and the "first generic market exclusivity period," as well as the deepening understanding of drug patents by domestic pharmaceutical companies, domestic companies are not only accelerating their布局 in innovative drugs but also increasingly competing for first generic drugs that offer market and pricing advantages. This is especially true for blockbuster varieties with global sales exceeding US$1 billion.