



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2022-11-04
Posting Date:2022-11-04 Views:
Views: On November 1 and 2, the official website of the National Medical Products Administration successively issued announcements on revising the package inserts for several injection products, involving products such as Astragalus Polysaccharide for Injection, Breviscapine Sodium Chloride Injection, Banlangen (Isatis root) Injection, and Liver Hydrolysate Injection.
In recent years, with the increasingly strong social demand for improved drug quality and the stringent requirements of centralized procurement for drug quality and supply, efforts such as post-marketing reevaluation of injectable products and the generic drug consistency evaluation have been deeply advanced. The clinical use of injections has been standardized, further ensuring the safety and accessibility of medication for the public.
Coincidentally, the Jiangxi Provincial Pharmaceutical Procurement Service Platform recently released the "Announcement on Soliciting Opinions on the Inter-Provincial Alliance Centralized Volume-Based Procurement Document (Draft)" directly targeting drugs that have not passed consistency evaluation, with a particular focus on injection products.
As early as 2019, the national "Notice on Several Policy Measures for Further Deepening the Reform of the Medical and Health System Using Centralized Drug Procurement and Utilization as a Breakthrough" mentioned the need to build a national drug public procurement market and a multi-party linkage procurement pattern. "For drugs not included in the national centralized procurement and utilization scope, local areas can learn from the experience of national centralized drug procurement and utilization."
Guided by this document, the forms of volume-based procurement have become increasingly diverse. In addition to national centralized procurement, provincial procurement, and inter-provincial alliances, more flexible models such as special volume-based procurement have emerged. Centralized procurement for varieties that have not passed consistency evaluation can also be seen as a manifestation of the flexibility of volume-based procurement.
Industry experts point out that with the advancement of the generic drug consistency evaluation policy and the active布局 of domestic pharmaceutical companies, the number of drugs passing the evaluation is constantly increasing. For enterprises, to increase product volume, enhancing their own competitiveness is the key! Although various regions have already paid attention to centralized procurement of drugs that have not passed consistency evaluation, in the long-term trend, passing the consistency evaluation remains a strategic choice to seize the major market share.
15 Major Clinical Varieties Included
Injectables Become the Focus of Centralized Procurement
In fact, there are precedents for centralized procurement of drugs that have not passed the evaluation, and Jiangxi Province is not carrying out such procurement for the first time.
As early as November 2019, the Jiangxi Provincial People's Government issued the "Notice on the Implementation Plan for Centralized Drug Volume-Based Procurement and Utilization in Jiangxi Province," announcing that it would select some drugs with large clinical usage, high procurement amounts, and relatively sufficient competition from among the generic drugs corresponding to the generic names of drugs that have not passed the quality and efficacy consistency evaluation, to organize and implement volume-based procurement.
This inter-provincial alliance procurement initiated by Jiangxi is already the third round of centralized procurement in Jiangxi Province.
In the 2019 Jiangxi Provincial Drug Volume-Based Procurement project, procurement was carried out for varieties outside the national and provincial volume-based procurement scope. In the second round of procurement for drugs that had not passed the evaluation, the scope expanded from a single province to an alliance format, involving four provinces: Jiangxi, Guangdong, Henan, and Hubei. This current procurement has further expanded to a 15-province alliance, covering a wider range, including most areas of the Northwest, Northeast, and other regions.
Judging from the procurement documents of this 15-province alliance led by Jiangxi, this procurement continues the overall approach of the first and second rounds, with no major changes.
According to the procurement documents, the procurement list includes 15 varieties such as papaverine, with the procurement cycle set for two years in principle.
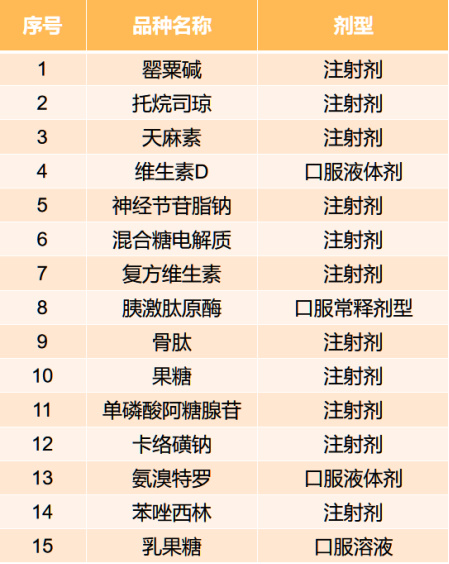
All 15 varieties in this procurement are chemical drugs with relatively large clinical usage and high procurement amounts. In terms of dosage form, injections are the most numerous, with 11 varieties, accounting for over 70%. In terms of therapeutic area, drugs for the digestive tract and metabolism are the most numerous, with 5 types, while no other therapeutic area exceeds 2 types.
Among them, lactulose, oxacillin, and pancreatic kininogenase also appeared in Jiangxi's second round of drug procurement list.
Lactulose promotes defecation and is commonly used clinically to treat chronic or habitual constipation. Data shows that sales of lactulose oral solution in China's three major terminals and six major markets exceeded 1.8 billion yuan in 2021, a year-on-year increase of 23.08%. In the ranking of chemical drugs for treating constipation, lactulose oral solution firmly ranks first. Over a dozen domestic companies hold production approvals for lactulose oral solution. Currently, multiple companies have submitted marketing applications for this product under the new registration classification, among which Sichuan Defeng Pharmaceutical's product has been successfully approved for production and deemed to have passed the consistency evaluation, being the first domestic company to do so.
Oxacillin Sodium for Injection is a penicillin antibiotic. It was included in the first batch of the National Essential Drugs List as early as 2009. Data shows that there are 20 domestic manufacturers of this product, including well-known pharmaceutical companies such as Kelun Pharmaceutical, North China Pharmaceutical, and CSPC Zhongnuo Pharmaceutical, with no foreign varieties.
Regarding procurement volume, the rules are the same as the previous two rounds of procurement. The agreed procurement volume for the first year for each variety is determined as follows:
If one company wins the bid, 50% of the baseline agreed procurement volume for the first year is taken as the agreed procurement volume for the first year.
If two companies win the bid, 75% of the baseline agreed procurement volume for the first year is taken as the agreed procurement volume for the first year, of which 50% is allocated as the first-year agreed procurement volume for the company with the lowest bid price, and 25% is allocated as the first-year agreed procurement volume for the company with the highest comprehensive score.
This procurement still adopts the "dual envelope" model for comprehensive evaluation, with the weight of economic and technical standards and commercial bids at 6:4. The product of the company with the lowest bid price and the product of the company with the highest comprehensive evaluation score are proposed to win; if the company with the lowest bid price and the highest comprehensive score is the same, only one company wins.
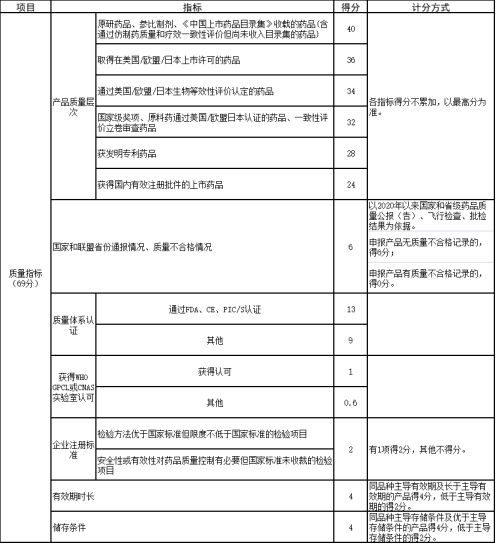
The score for economic and technical standards is generally composed of the following aspects:
Quality Indicators (69 points): Mainly the product quality level, divided into 6 aspects including originator, passed consistency evaluation, marketed in Europe, the US, or Japan, etc. Scores are not cumulative, and the highest score among them is taken.
Supply Guarantee Indicators (13 points): Assigned scores based on different distribution rate indicators.
Comprehensive Indicators (18 points): Includes several indicators such as procurement失信 records, national centralized procurement selection status, market share, medical institution coverage, industry ranking, etc., with different scores.
Additionally, the commercial bid has a full score of 100 points, of which price reduction accounts for 50 points and the absolute bid price accounts for 50 points.
From the procurement rules, it can be seen that this Jiangxi alliance procurement not only emphasizes the balance between technology and price but also places more emphasis on the scoring role of technical standards.
"Under the 'dual envelope' model, in addition to controlling product prices, factors such as a company's own product advantages and solid technical indicators are also particularly important!" Industry analysts pointed out that from the weight of evaluation indicators, non-price factors such as product quality and supply guarantee account for a relatively high proportion, reflecting the simultaneous requirements for drug quality and price. Furthermore, comprehensive indicators such as market share and industry ranking are more favorable to large-scale enterprises.
"Price War" Unavoidable
Passing Evaluation Remains the Primary Task
According to the work deployment of the National Healthcare Security Administration, drugs that have passed the consistency evaluation are subject to centralized procurement organized by the state. Currently, as a supplement to the mainstream volume-based procurement model, volume-based procurement for drugs that have not passed the evaluation is also receiving increasing regulatory attention.
On June 5, 2020, nine ministries and commissions, including the National Health Commission, jointly issued the "Notice on Issuing the Key Points for Correcting Malpractice in the Field of Pharmaceutical Purchasing and Sales and Medical Services in 2020," which explicitly proposed to "promote pilot programs for volume-based procurement of non-evaluated drugs and high-value medical consumables in various regions."
On February 26, 2020, the Fujian Provincial Healthcare Security Bureau announced the "Fujian Provincial Centralized Drug Volume-Based Procurement Document," officially announcing the 14 procurement varieties and corresponding procurement volumes for this round. These 14 varieties were selected from drugs with large procurement amounts, sufficient competition, mature clinical use, and no generic drugs under the same generic name that had passed the consistency evaluation. They included drugs such as esomeprazole, moxifloxacin, and leuprorelin. Most of these drugs are injections, with a procurement cycle of one year from the official implementation of the centralized volume-based procurement.
On December 11, 2019, the Hebei Provincial Centralized Procurement Center for Medical Drugs and Devices conducted centralized procurement for 15 types of outpatient drugs for hypertension and diabetes. The results showed that 13 drugs entered the proposed selection list, among which some varieties were selected by pharmaceutical companies that had not passed the consistency evaluation.
On August 3, 2019, the "Opinions on Strengthening the Centralized Procurement of Drugs and Medical Consumables" issued by the Shandong Provincial Healthcare Security Bureau also mentioned that the varieties for procurement would focus on drugs that have not passed the quality and efficacy consistency evaluation.
…
Under the generic drug consistency evaluation mechanism, several provinces and cities have previously specified that "if three or more manufacturers of a drug variety pass the consistency evaluation, drugs that have not passed the consistency evaluation will no longer be selected in centralized drug procurement." A large number of drugs that have not passed the evaluation have been suspended from procurement.
In December 2021, the state issued the "14th Five-Year Plan for National Drug Safety and High-Quality Development," clarifying the need to further implement the requirements of the review and approval system reform and continuously promote the consistency evaluation of chemical generic drugs.
In fact, taking this Jiangxi provincial alliance procurement as an example, the product quality indicators not only account for the highest proportion of scores but also include multiple scoring indicators designed from different dimensions.
Passing the consistency evaluation means that the quality and efficacy of the generic drug have been recognized, and drugs that have passed the evaluation are linked to national centralized procurement. This is of great significance for drug certificate holders, drug manufacturers, clinical practice, and public medication. It also helps promote the export of domestic generic drugs.
On the other hand, with the continuous deepening of the drug review and approval system reform, drug quality control will become increasingly stringent. Generic drugs that have not passed the consistency evaluation will face greater restrictions and more intense competition, ultimately having to be suspended from centralized procurement, having their drug approvals revoked, or even facing market withdrawal.
From this perspective, accelerating the consistency evaluation will still be a necessary choice for most pharmaceutical companies. Industry insiders remind that according to policy requirements, once three manufacturers (including the originator and generics that have passed the consistency evaluation) exist for a drug, it can be included in national centralized procurement. Being the first to pass the evaluation is of great significance for enterprises, as it helps them further expand their market. Companies with large-volume drugs that have not passed the evaluation should accelerate their efforts to pass the evaluation as soon as possible!