



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2022-10-14
Posting Date:2022-10-14 Views:
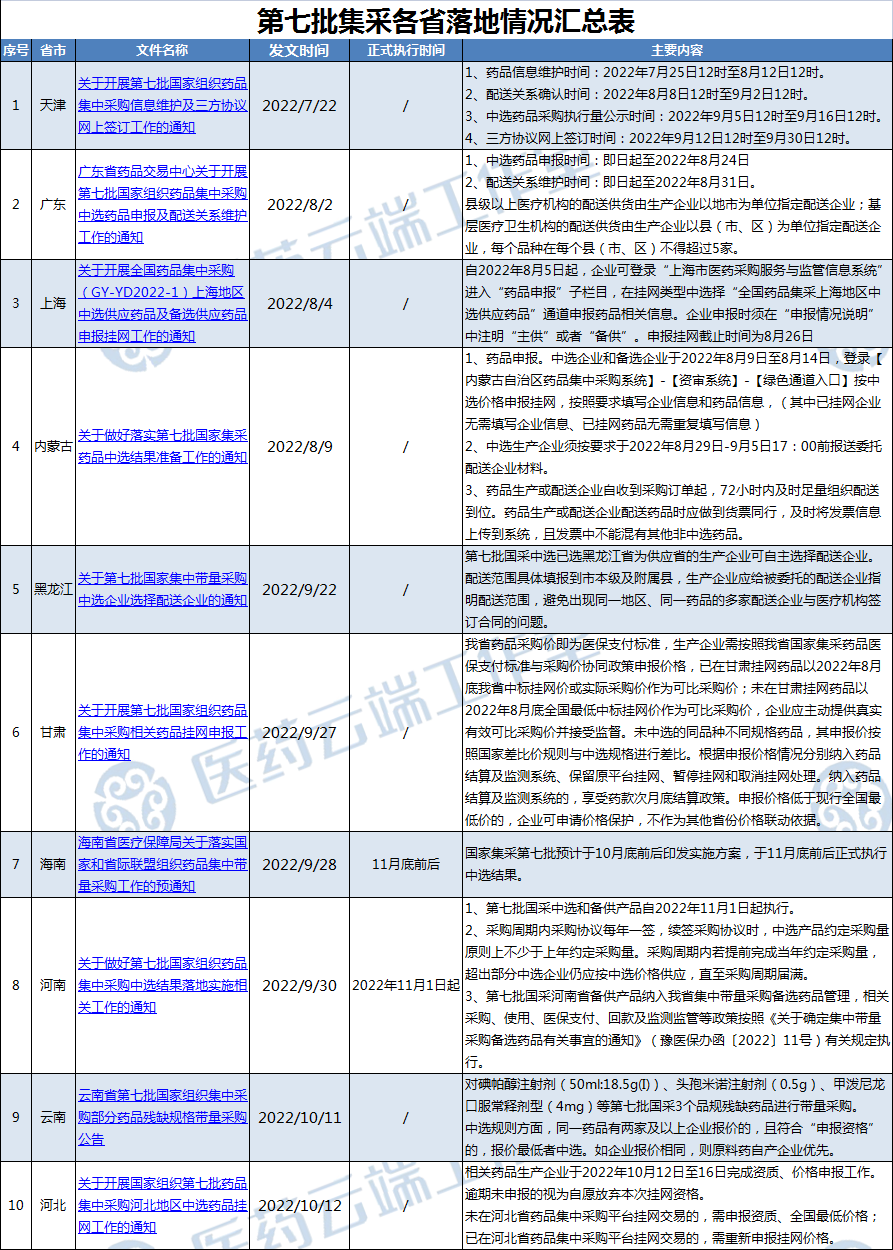
Views: The National Joint Procurement Office previously notified that the selected results of the seventh round of national centralized procurement will be implemented in November 2022, with the specific execution date subject to the notices issued by each region.
Henan and Hainan have issued documents, specifying that the seventh round of national centralized procurement will be implemented on November 1, 2022, and by the end of November, respectively. Yunnan has initiated volume-based procurement for three incomplete specifications, and Hebei has explicitly required the completion of qualification and price declaration work from October 12 to 16.
On July 18, the National Joint Procurement Office issued a notice regarding the selected results of the seventh round of national centralized procurement.
The announcement stated that the selected results of this round will be implemented in November 2022, with the specific execution date subject to the notices issued by each region. A total of 60 drugs were successfully procured in this round, with an average price reduction of 48% for the selected drugs, covering 31 therapeutic categories. These include not only common and chronic disease medications such as those for hypertension, diabetes, anti-infection, and gastrointestinal diseases, but also drugs for major diseases such as lung cancer, liver cancer, kidney cancer, and colorectal cancer. Based on the agreed procurement volume, it is estimated that annual cost savings could reach 18.5 billion yuan.
Currently, various provinces are successively issuing notices regarding the implementation of the seventh round of national centralized procurement. The summary is as follows:
Yunnan: Volume-Based Procurement for Three Incomplete Specifications, Lowest Bidder Wins
On October 11, Yunnan Province issued the "Announcement on Volume-Based Procurement for Incomplete Specifications of Some Drugs from the Seventh Round of National Centralized Procurement in Yunnan Province," initiating volume-based procurement for three incomplete specifications from the seventh round: Iopamidol Injection (50ml: 18.5g (I)), Cefminox Injection (0.5g), and Methylprednisolone Oral Immediate-Release Formulation (4mg).
The total estimated procurement volume for the three specifications is 3.8283 million (tablets/capsules/packs/vials). The procurement entities are medical institutions across the province and designated medical insurance pharmacies. The procurement cycle is generally one year.
Regarding pricing: For varieties that were selected in the national centralized procurement but did not choose to supply the Yunnan region, the declared price must not exceed the national selected price. For varieties that meet the drug declaration qualifications but do not have a national centralized procurement selected price, the declared price must not exceed the selected drug price in Yunnan Province (different specifications are converted based on the specification price difference ratio of the selected drug).
Regarding the selection rules: If two or more companies submit bids for the same drug and meet the "declaration qualifications," the company offering the lowest price wins. If the bid prices are the same, the company that produces its own active pharmaceutical ingredient (API) will have priority.
Selected drugs will be listed in the volume-based procurement section of the Yunnan Provincial Drug Centralized Procurement Platform. They will be subject to relevant national policies such as advance payment from the centralized procurement fund and retention of savings. The selected companies may select no more than three distribution companies on the Yunnan Provincial Drug Centralized Procurement Platform for distribution and establish distribution relationships.
If a selected variety is suspended from production, sale, use, or import by the drug regulatory authority, its selection qualification will be revoked.
Hebei: Complete Qualification and Price Declaration Work from October 12 to 16
On October 12, the Hebei Provincial Centralized Procurement Center for Medical Drugs and Devices issued the "Notice on Carrying Out the Online Listing of Selected Drugs from the Seventh Round of National Centralized Drug Procurement in Hebei Region," requiring relevant drug manufacturers to complete qualification and price declaration work from October 12 to 16, 2022. Failure to declare within the time limit will be deemed as voluntarily giving up the qualification for online listing.
Selected drugs and alternative drugs for the seventh round of national centralized procurement in the Hebei region must be declared. The declared drug specifications and packaging must fall within the scope of the "National Drug Centralized Procurement Document (GY-YD2022-1)." Selected drugs from other regions, as well as non-selected drugs of the same variety, that intend to be listed for sale in Hebei Province may also declare and be listed according to Hebei Province policies.
For drugs not yet traded on the Hebei Provincial Drug Centralized Procurement Platform, qualification information and the national minimum price must be declared. For drugs already traded on the Hebei Provincial Drug Centralized Procurement Platform, the listed price must be re-declared.