



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2021-10-12
Posting Date:2021-10-12 Views:
Views: Gilead Sciences recently announced that Health Canada has approved its targeted anticancer drug Trodelvy (sacituzumab govitecan-hziy) through Project Orbis for the treatment of adult patients with metastatic triple-negative breast cancer (mTNBC). Specifically, the drug is indicated for adult patients with unresectable locally advanced or metastatic TNBC who have previously received at least two prior therapies, with at least one of those therapies administered for metastatic disease. Trodelvy is a novel, first-in-class antibody-drug conjugate (ADC) targeting Trop-2, a cell surface protein overexpressed in TNBC and other solid tumors.
Notably, Trodelvy is the first therapy to demonstrate superiority over standard of care in treating metastatic TNBC, marking a significant advancement in TNBC treatment. In the Phase 3 ASCENT study, compared with chemotherapy, Trodelvy significantly prolonged progression-free survival (median PFS: 4.8 months vs 1.7 months) and reduced the risk of disease progression or death by 57% (HR=0.43, p<0.0001), regardless of brain metastasis status. Additionally, compared with chemotherapy, Trodelvy significantly extended overall survival (median OS: 11.8 months vs 6.9 months) and reduced the risk of death by 49% (HR=0.51, p<0.0001).
This approval in Canada also marks the fifth approval for Trodelvy through Project Orbis. Previously, Trodelvy received approvals in the United States, Switzerland, the United Kingdom, and Australia through this initiative. Project Orbis was launched by the FDA Oncology Center of Excellence (OCE) in September 2019 to provide a collaborative framework for concurrent submission and review of oncology products between the FDA and its international regulatory partners, aiming to expedite the review process and synchronize approvals of cancer treatments across different countries. In addition to the Project Orbis approvals, the European Medicines Agency (EMA) accepted the Marketing Authorization Application (MAA) for Trodelvy in March of this year. Furthermore, through licensing partner Everest Medicines, Trodelvy is also undergoing regulatory review in China and Singapore.
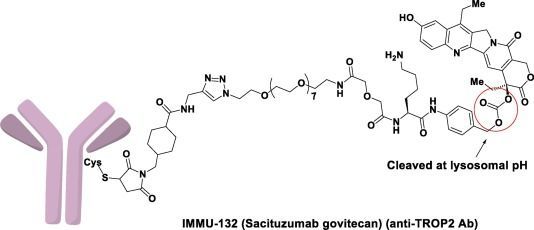
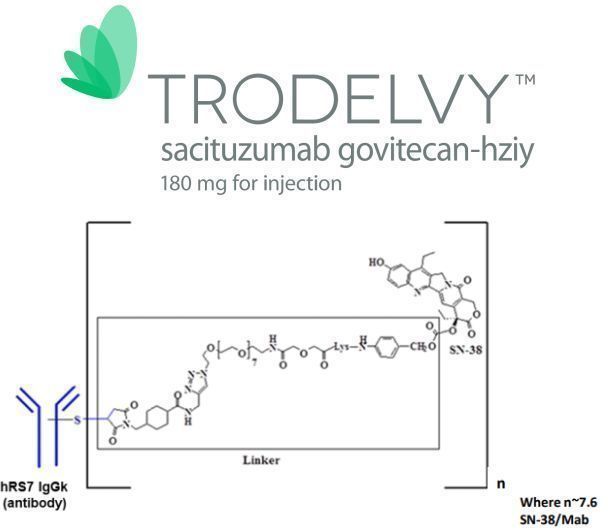
Structural features of Trodelvy (Image source: broadpharm.com)
Trodelvy is a novel, first-in-class antibody-drug conjugate (ADC) composed of a humanized IgG1 antibody targeting the Trop-2 antigen conjugated with SN-38, the active metabolite of the chemotherapeutic agent irinotecan (a topoisomerase I inhibitor), with a drug-to-antibody ratio of 7.6:1. Trop-2 is a cell surface protein frequently expressed in many epithelial tumors, including TNBC, and is expressed in over 90% of TNBC cases. Trodelvy binds to Trop-2 and delivers the anticancer agent SN-38 to kill cancer cells.
Trodelvy was developed by Immunomedics, whose proprietary ADC platform centers on the use of a novel linker that does not require enzymatic cleavage to release the payload. This linker enables the delivery of active drug both within tumor cells and in the tumor microenvironment, producing a bystander effect. In April 2019, Everest Medicines entered into an agreement with Immunomedics to obtain rights to Trodelvy in Greater China, South Korea, Mongolia, and Southeast Asian countries. In September 2020, Gilead acquired Immunomedics for $21 billion, bringing Trodelvy into its portfolio.
.
In the United States, Trodelvy has been approved for two indications: (1) for the treatment of adult patients with unresectable locally advanced or metastatic TNBC who have previously received at least two prior therapies, with at least one of those therapies administered for metastatic disease; and (2) for the treatment of adult patients with locally advanced or metastatic urothelial cancer (UC) who have previously received platinum-containing chemotherapy and either a PD-1 inhibitor or a PD-L1 inhibitor.
In China, Trodelvy (sacituzumab govitecan) was included in the 2020 edition of the Guidelines for Standard Diagnosis and Treatment of Advanced Breast Cancer in China in October 2020. This guideline was jointly developed by the Breast Cancer Expert Committee of the National Cancer Quality Control Center, the China Anti-Cancer Association Breast Cancer Committee, and the China Anti-Cancer Association Committee on Clinical Research of Anticancer Drugs. In May 2021, China's National Medical Products Administration (NMPA) accepted the biologics license application for Trodelvy and granted it priority review.
Breast cancer is the most common type of cancer among women, with over 2 million new cases diagnosed globally each year. Triple-negative breast cancer (TNBC) accounts for approximately 15% of all breast cancers and is more common in women under 50 compared to other types of breast cancer. TNBC is defined by the absence of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER-2) expression. It progresses rapidly and has a poor prognosis, with a five-year survival rate of less than 15%. TNBC does not respond to hormone therapy or HER2-targeted therapies (such as Roche's Herceptin), leaving very limited clinical treatment options and making chemotherapy the mainstay of treatment.
The regulatory approval of Trodelvy was based on results from the Phase 3 ASCENT (NCT02574455) trial, an international, open-label Phase III study that enrolled more than 500 patients with mTNBC who had received at least two prior therapies for metastatic disease. Patients were randomly assigned to receive either Trodelvy or chemotherapy chosen by the physician. The primary endpoint was progression-free survival (PFS), with secondary endpoints including overall survival (OS), objective response rate (ORR), duration of response (DOR), time to response, safety, and tolerability.
Results showed that the study met its primary and key secondary endpoints. The Trodelvy group demonstrated a statistically significant improvement in PFS compared with the chemotherapy group (median PFS: 4.7 months vs 1.7 months), with a 57% reduction in the risk of disease progression (HR=0.43, 95% CI: 0.35-0.54, p<0.0001). The study also met its key secondary endpoint, showing a statistically significant improvement in OS for the Trodelvy group compared with the chemotherapy group (median OS: 11.8 months vs 6.9 months), with a 49% reduction in the risk of death (HR=0.51; 95% CI: 0.41-0.62; p<0.0001). In addition, the ORR was significantly higher in the Trodelvy group compared with the chemotherapy group (35% vs 5%).
Original source: Gilead Marks Fifth Approval for Trodelvy in Metastatic Triple-Negative Breast Cancer Under Project Orbis Initiative with Health Canada Authorization