



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2021-09-30
Posting Date:2021-09-30 Views:
Views: On September 27, ChemoCentryx, Vifor Pharma, and Kissei Pharmaceutical jointly announced that Japan's Ministry of Health, Labour and Welfare (MHLW) has approved Tavneos (avacopan), a first-in-class oral small-molecule complement C5a receptor inhibitor, for marketing. It is indicated for the treatment of the two main types of anti-neutrophil cytoplasmic autoantibody (ANCA)-associated vasculitis: microscopic polyangiitis (MPA) and granulomatosis with polyangiitis (GPA). This marks the first global approval for this C5aR inhibitor.
ANCA-associated vasculitis is a systemic disease in which excessive activation of the complement pathway further activates neutrophils, leading to inflammation and destruction of small blood vessels, as well as organ damage and failure, particularly in the kidneys. Current treatments for this condition include non-specific immunosuppressive therapies (such as cyclophosphamide or rituximab), along with long-term daily use of glucocorticoids (steroids), which may pose significant clinical risks, including death from infection.
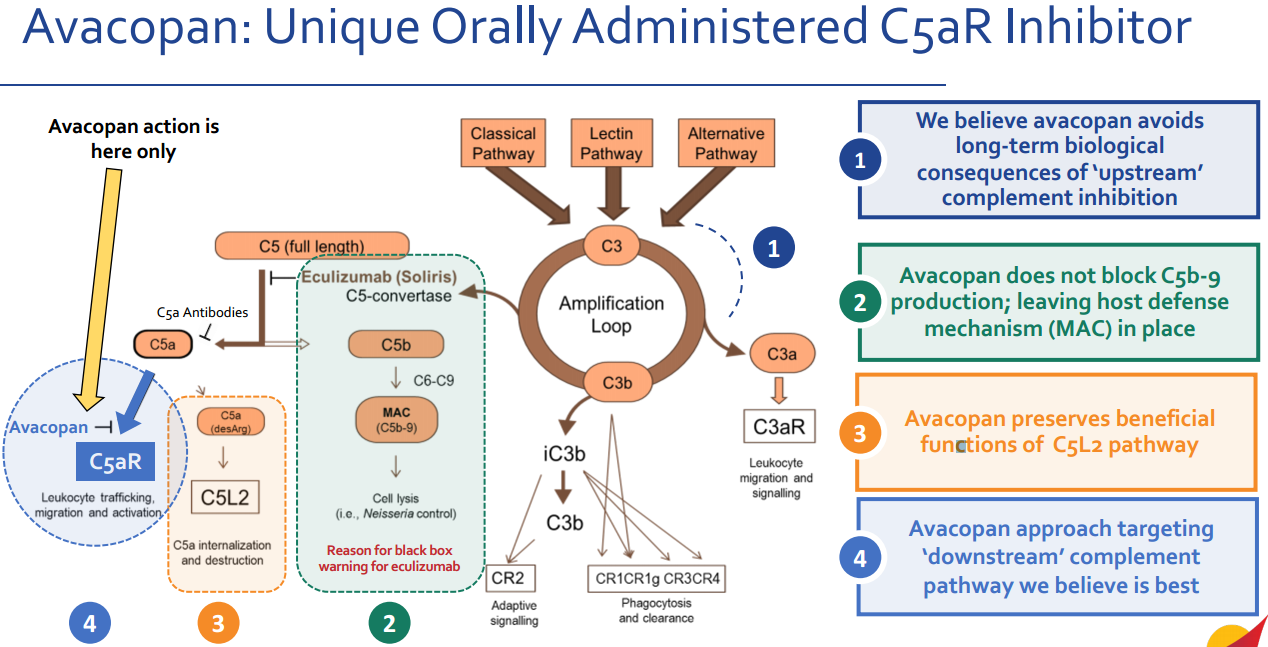
Avacopan employs a novel, highly targeted mechanism of action in the treatment of ANCA-associated vasculitis and other complement-driven autoimmune and inflammatory diseases. It avoids upstream complement inhibition and does not block C5b-9 formation, thereby preserving the body's defense mechanisms (membrane attack complex, MAC), while also retaining the beneficial functions of the C5L2 pathway.
On July 6 of this year, ChemoCentryx announced that the PDUFA target date for the U.S. new drug application (NDA) of avacopan had been extended to October 7, 2021. The original PDUFA date for the avacopan NDA was July 7, 2021.