



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2021-09-06
Posting Date:2021-09-06 Views:
Views: On September 3, the General Office of the National Health Commission issued the "Procedures for Adjusting the National Key Monitoring Rational Drug Use List," consisting of ten provisions that clarify the selection scope, adjustment principles, cycles, and work procedures for key monitoring rational drug use.
Among them, the update and adjustment cycle for the list shall generally be no less than 3 years, with 30 drugs typically included. Provincial health administrative departments shall, based on the 30 drugs submitted by their respective provinces, incorporate the new version of the list to form a provincial key monitoring rational drug use list, which shall be promptly reported to the National Health Commission for record.
The adjustment process includes four stages: initiation of adjustment, local selection and recommendation, expert aggregation, and announcement of results.
For drugs removed from the previous list, local health administrative departments shall continue monitoring for at least one year.
Notably, the drugs included in this list are primarily chemical drugs and biologics, with no mention of traditional Chinese patent medicines.
The first National Key Monitoring Rational Drug Use List dates back to July 1, 2019, when the Medical Administration Bureau of the National Health Commission issued the "Notice on Printing and Distributing the First Batch of National Key Monitoring Rational Drug Use List (Chemical Drugs and Biologics)." Industry interpretation viewed this as targeting "adjuvant drugs."
The first batch mainly included gangliosides, porcine cerebroside, oxiracetam, creatine phosphate sodium, deproteinized calf serum, alprostadil, troxerutin and cerebroprotein hydrolysate, coenzyme complex, salvia miltiorrhiza and ligustrazine, invert sugar and electrolytes, mouse nerve growth factor, thymopentin, ribonucleic acid II, edaravone, ossotide, cerebroprotein hydrolysate, ribonucleic acid, vinpocetine, deproteinized calf blood extract, and cinepazide maleate.
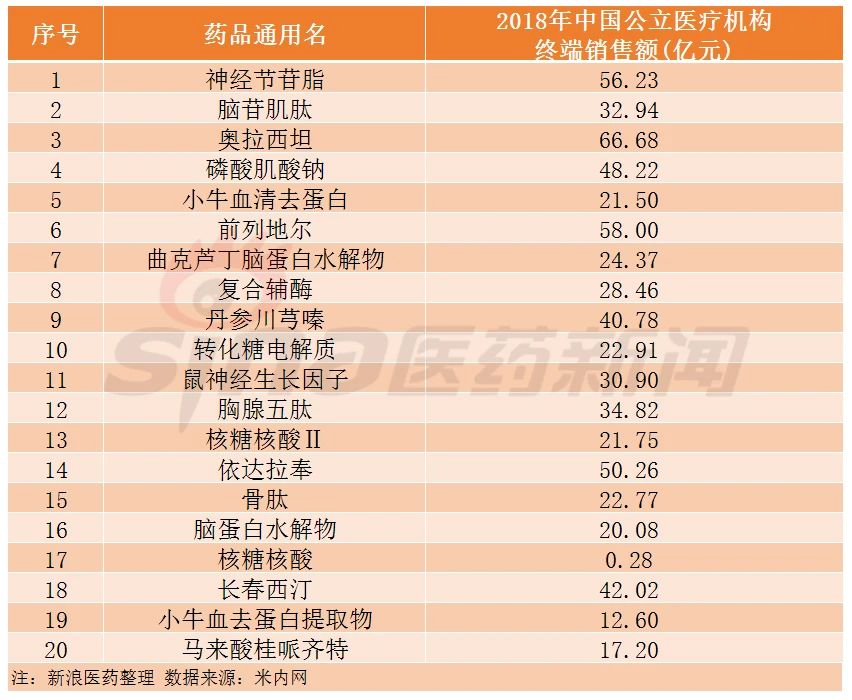
According to previous statistics by Sinayiyao, the 20 key monitoring drugs involve more than 200 companies, including over 20 listed companies, such as Sihuan Pharmaceutical, Fosun Pharma, Livzon Group, Kunming Pharmaceutical Group, Saisheng Pharmaceutical, and Buchang Pharmaceutical.
In addition, various provinces and cities have successively introduced local key monitoring drug lists. According to incomplete statistics, in addition to the 20 national-level drugs, provincial, municipal, and even medical institutions have implemented key monitoring for certain drugs, covering over 200 drugs including chemical drugs, biologics, and traditional Chinese patent medicines.
Appendix:
Procedures for Adjusting the National Key Monitoring Rational Drug Use List
Article 1 These procedures are formulated to strengthen the management of clinical rational drug use in China, promote a more scientific and reasonable formulation and adjustment of the National Key Monitoring Rational Drug Use List (hereinafter referred to as the List), continuously standardize clinical medication practices, and safeguard the health rights and interests of the people.
Article 2 Drugs included in the List shall be chemical drugs and biologics with prominent issues in clinical irrational use, abnormally high usage expenditure, and significant impact on medication rationality. These mainly include adjuvant drugs, antineoplastic drugs, antimicrobial drugs, proton pump inhibitors, glucocorticoids, parenteral nutrition drugs, etc.
Article 3 The adjustment of the List adheres to the principles of "openness and transparency, local recommendation, and dynamic adjustment," with the goal of standardizing clinical medication practices and promoting rational drug use.
Article 4 The update and adjustment cycle for the List shall generally be no less than 3 years, and the number of drugs included in the List shall typically be 30.
Article 5 The National Pharmaceutical Affairs and Pharmacotherapy Committee (hereinafter referred to as the National Pharmaceutical Affairs Committee) shall provide professional technical support for the scientific adjustment of the List and undertake the aggregation, organization, and analysis of materials recommended by local authorities.
Article 6 The adjustment of the List consists of four stages: initiation of adjustment, local selection and recommendation, expert aggregation, and announcement of results.
(1) Initiation of adjustment. The National Health Commission issues a notice on the adjustment of the List, specifying the timeline, materials to be submitted, work requirements, etc.
(2) Local selection and recommendation. Secondary and above general hospitals, based on comprehensive factors such as the current status of clinical irrational drug use, usage expenditure, and clinical value, after deliberation and selection by their hospital's Pharmaceutical Affairs and Pharmacotherapy Committee, shall rank drugs by generic name (without dosage form distinction) in descending order of recommendation strength, and submit information on the top 30 recommended drugs to the provincial health administrative department with the hospital's official seal.
Provincial health administrative departments shall assign corresponding scores to the 30 drugs recommended by each hospital, i.e., the drug ranked first receives 30 points, the second receives 29 points, and so on, with the 30th ranked drug receiving 1 point. Based on the generic name, provincial health administrative departments shall sum the scores of all drugs submitted by secondary and above general hospitals within their jurisdiction to obtain the total recommended score for each drug, and then rank them in descending order (drugs with the same score may be ranked in parallel and occupy subsequent ranks). Information on the top 30 drugs (including other drugs tied for 30th place) shall be submitted to the Medical Administration Bureau of the National Health Commission with the official seal.
(3) Expert aggregation. The National Health Commission entrusts the National Pharmaceutical Affairs Committee to conduct formal reviews of the materials submitted by local authorities, using the same calculation method as the provincial health administrative departments to determine the top 30 drugs.
(4) Announcement of results. The National Health Commission announces the adjustment results, releases the new version of the List, and proposes management requirements.
Article 7 Provincial health administrative departments shall, based on the 30 drugs submitted by their respective provinces, incorporate the new version of the List to form a provincial key monitoring rational drug use list, which shall be promptly reported to the National Health Commission for record. Medical institutions at all levels shall, based on the provincial list and combined with their actual medication practices, form their own institutional key monitoring rational drug use list. The provincial and institutional key monitoring rational drug use lists shall be made public to society in accordance with requirements through government affairs disclosure, hospital affairs disclosure, official website announcements, etc.
Article 8 Local health administrative departments shall attach great importance to strengthening pharmaceutical affairs management in medical institutions, effectively enhance organizational leadership and coordination, and ensure the implementation of rational drug use work. They shall formulate and improve clinical application guidelines for relevant drugs, clarify medication guidelines and administration regimens, and fully utilize modern information tools to strengthen rational drug use supervision.
Article 9 For drugs removed from the previous list, local health administrative departments shall continue monitoring for at least one year, tracking information such as prescription review, usage volume, and usage expenditure, to promote continuous improvement in clinical rational drug use.
Article 10 These procedures shall take effect from the date of issuance.
END