



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2021-08-27
Posting Date:2021-08-27 Views:
Views: The battle for the $6 billion oseltamivir market has never stopped.
01
CSPC Makes Its Move
On August 19, Insight Database showed that the generic version of oseltamivir capsules from CSPC Pharmaceutical Group had entered the administrative approval stage and was expected to be approved soon.
CSPC's presence in the anti-influenza drug market is not recent. Back in 2009, at the "Expert Seminar on TCM Diagnosis and Treatment of Influenza A (H1N1)" held in Beijing, CSPC Ouyi's Qingre Jiedu Soft Capsules were recommended by attending experts for the treatment of H1N1 influenza.
Previously, CSPC Ouyi also submitted and received acceptance for a generic version of baloxavir marboxil tablets as a Class 4 generic drug. This is the first generic version of this drug to be filed in China. Baloxavir marboxil is another major anti-influenza drug from Roche following Tamiflu (oseltamivir). It directly inhibits viral replication and is used to treat oseltamivir-resistant strains and avian influenza virus strains.
According to the "2021-2025 Anti-Influenza Drug Market In-depth Research and Investment Forecast Analysis Report" released by the Xinjie Industry Research Center, global sales of major anti-influenza drugs have been increasing year by year with a relatively fast growth rate. In 2019, global anti-influenza drug sales reached approximately $1.8 billion, and are expected to reach $3 billion by 2024. Oseltamivir and baloxavir marboxil are the top-selling drugs in this category.
In recent years, the number of influenza cases in China has been rising exponentially. The above report points out that in 2019, there were over 9 million influenza cases in China. As patient numbers grow, the scale of China's anti-influenza drug market continues to expand.
Data from Menet show that in 2019, sales of cough and cold medications in China's urban public hospitals, county public hospitals, urban community centers, and township health centers (collectively referred to as public medical institutions) reached 18.1 billion yuan, a year-on-year increase of 4.64%.
According to CSPC's 2020 annual report, compared with 2019, CSPC's total revenue in 2020 increased by 12.8% to 24.942 billion yuan, and sales revenue increased by 13.8% to 20.405 billion yuan. Its main product, arbidol hydrochloride tablets, used for antiviral treatment and influenza prevention, was included in multiple editions of COVID-19 diagnosis and treatment guidelines in 2020.
02
The Oseltamivir Arena
In the arena of the $6 billion oseltamivir market, competition among pharmaceutical companies has never ceased.
The originator of oseltamivir is Roche, marketed under the brand name Tamiflu. Around 2006, Roche licensed oseltamivir to Shanghai Zhongxi Pharmaceutical and Dongyangguang Pharmaceutical, which marketed the drug under the brand names "Erfei" and "Kewei," respectively. Since Shanghai Zhongxi's version did not achieve significant market circulation, Dongyangguang Pharmaceutical has remained the dominant player in China's oseltamivir market.
Currently, oseltamivir is mainly available in capsule and granule formulations. Pending applications also include dry suspension and orally disintegrating tablets. A query on the Insight WeChat mini-program revealed 32 product listings for oseltamivir, with 15 domestic companies having filed generic applications for oseltamivir capsules. Several applications have already been accepted by the CDE.
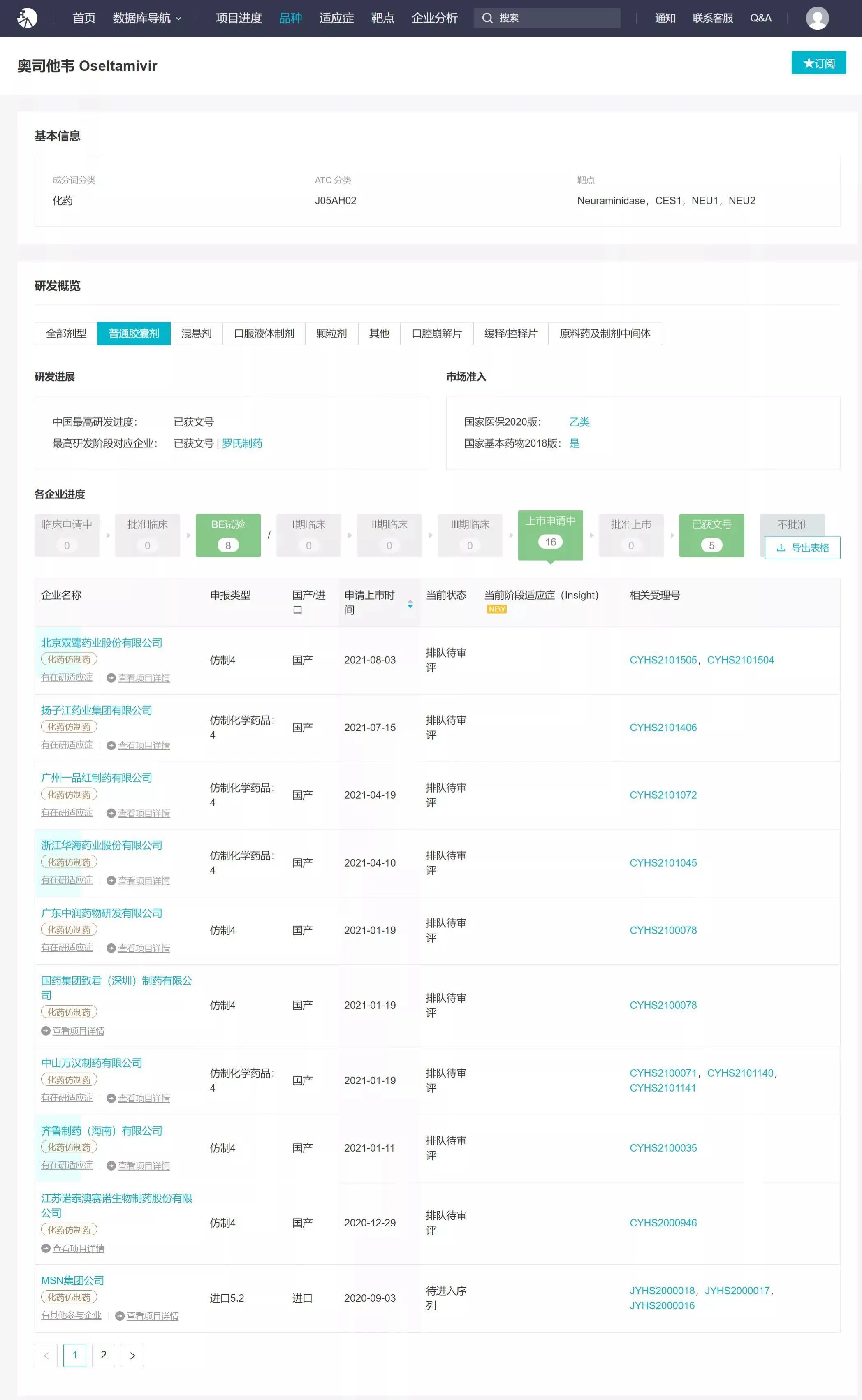
Image source: Insight Database
BrightGene Pharmaceutical
On July 30, the National Medical Products Administration (NMPA) website announced that BrightGene Pharmaceutical's generic oseltamivir phosphate capsules had been approved, making it the first generic version approved in China.
Beijing Star Island Pharmaceutical
On September 27, Beijing Star Island Pharmaceutical's Class 2.2 improved new drug application for "oseltamivir phosphate orally disintegrating tablets" was accepted by the CDE. This was the first improved new drug application for oseltamivir submitted by a domestic company.
Kelun
On July 31, Kelun Pharmaceutical's Class 4 generic application for "oseltamivir capsules" was accepted by the CDE.
Yangtze River Pharmaceutical
On July 31, Insight Database showed that Yangtze River Pharmaceutical Group Beijing Haiyan Pharmaceutical Co., Ltd. had initiated a bioequivalence study for "oseltamivir phosphate granules" at a 25 mg strength.
Chengdu Beite
On April 2, the CDE website showed that Chengdu Beite's Class 4 generic application for oseltamivir phosphate capsules had been accepted.
In addition, the available strengths of oseltamivir are also expanding. A search on the NMPA website revealed that beyond the 75 mg capsule, Dongyangguang Pharmaceutical recently received approval for two exclusive strengths: 30 mg and 45 mg capsules. This will further meet patient market demand and expand Dongyangguang's sales reach.

03
A "Waterloo" for the Market Leader?
Oseltamivir, the undisputed leader in anti-influenza drugs, once propelled Dongyangguang Pharmaceutical to remarkable heights.
In 2018, oseltamivir was included in the "Influenza Diagnosis and Treatment Plan (2018 Edition)" as a recommended flu medication. Times make heroes, and the same holds true for oseltamivir.
Since oseltamivir was launched in 2006, there have been three major influenza outbreaks: the 2009 H1N1 swine flu, the 2016 H7N9 avian flu, and the 2017 H7N9 avian flu. Notably, in 2016 and 2017, Dongyangguang Pharmaceutical's revenue increased by approximately 36% and 70.11% year-on-year, respectively.
Its most enviable performance came in 2019, when Dongyangguang reported total revenue of 6.22 billion yuan, a 147.9% year-on-year increase. Among this, the Kewei series generated 5.934 billion yuan, a year-on-year increase of 163.42%, accounting for 95.33% of Dongyangguang's total revenue. Kewei granules brought in 4.273 billion yuan, representing 68.65% of total revenue, while Kewei capsules contributed 1.661 billion yuan, accounting for 26.68%.
However, under the impact of the global pandemic in 2020, this anti-influenza leader saw its relevance diminish. Dongyangguang's 2020 annual report showed sales of approximately 2.348 billion yuan, a 62.27% decrease compared with 2019. Sales of its core product, oseltamivir phosphate, were approximately 2.069 billion yuan (1.14784 billion yuan from granules and 920.89 million yuan from capsules), a 65.1% decline from the same period in 2019. Previously, oseltamivir phosphate, one of the largest single products in China's chemical drug market, accounted for over 90% of the company's total revenue.
Regarding the significant decline in oseltamivir phosphate's performance in 2020, Dongyangguang's annual report analyzed that the main reason was the impact of the global pandemic, which reduced domestic population mobility and heightened public health awareness, making people less susceptible to influenza than in previous years.
Notably, data from Menet shows that in the systemic antiviral drug sector, Dongyangguang offers not only oseltamivir but also famciclovir tablets, ganciclovir for injection, and valacyclovir hydrochloride tablets. In 2020, in addition to obtaining approval for the Class 1 new drug emitasvir phosphate capsules, it also received approval and passed the generic consistency evaluation for entecavir tablets. Its pending application for tenofovir alafenamide fumarate tablets is expected to be approved in 2021. The application for oseltamivir phosphate dry suspension, filed as a Class 3 generic in 2020, is currently under review.
Beyond its broad antiviral product line, Dongyangguang is also actively investing in innovative drugs and the insulin market. According to its annual report, R&D investment totaled over 1 billion yuan, accounting for 43.18% of its revenue, a year-on-year increase of 523.5%. Its insulin product development plan is comprehensive, covering both second-generation and third-generation insulin products. In the future, this 60 billion yuan insulin market may see new competitors emerge.
Dongyangguang Pharmaceutical once thrived on oseltamivir phosphate, but with a highly concentrated revenue stream, it also faces the risk of over-reliance on a single product. Although sales have declined recently, the company is actively diversifying its product lines to support future growth. Whether its future will be promising remains worth watching.