



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2021-06-18
Posting Date:2021-06-18 Views:
Views:
This year marks the 100th anniversary of the discovery of insulin, which revolutionized the care of patients with type 1 diabetes. Over the past century, scientists have gained a deeper understanding of the pathogenesis of different types of diabetes, leading to significant advances in patient care.
▋1922: Initial Discovery and First Use of Insulin
In 1921, Dr. Frederick Banting and his assistant Charles Best conducted a series of experiments investigating how the pancreas is involved in carbohydrate metabolism. They removed the pancreas from several dogs, causing them to develop symptoms of diabetes, and then injected pancreatic extracts into these dogs. The results showed that injection of pancreatic extract reduced blood glucose and urinary glucose levels in the dogs and improved their health. These findings indicated that pancreatic secretions are involved in carbohydrate metabolism.
Subsequently, together with Canadian chemist James Bertram Collip, they successfully purified insulin from pancreatic extracts and developed methods for large-scale production, making it available for patient use.
In 1922, 14-year-old type 1 diabetes patient Leonard Thompson received insulin injections and showed improvement in his symptoms. The researchers noted in their 1922 publication that these results unequivocally demonstrated the therapeutic value of pancreatic extracts for certain patients with type 1 diabetes!
This discovery spurred rapid progress in the use of insulin to treat type 1 diabetes. Dr. Banting and physiologist John Macleod were awarded the Nobel Prize in Physiology or Medicine in 1923 for their research on insulin.

▲Dr. Banting (right) and his assistant Mr. Best with a dog used in the experiments (Image source: NobelPrize.org, Credits: University of Toronto Archives)
▋1965: Histopathological Studies of the Pancreas in Diabetes
Although the discovery of insulin revolutionized the treatment of diabetes, the pathophysiology of the disease remained unclear for several decades. In 1965, a landmark histological study revealed that in patients with early-onset diabetes (what we now call type 1 diabetes), insulin-producing β cells were greatly reduced (to less than 10% of normal levels), and most patients with acute disease showed inflammatory infiltrates around and within the pancreatic islets. This suggested that β cells were under attack by "external factors."
These histological features differed from those of late-onset diabetes (type 2 diabetes), in which β cell volume typically ranged from 40-50% of normal.
These studies of patients with early-onset diabetes helped elucidate the pathological processes occurring in the pancreatic islets of diabetic patients, ultimately leading to new therapies that improved patient outcomes.
▋1978: Pioneering Studies on Diabetic Complications
By the mid-20th century, the association between diabetes and a range of degenerative conditions—including neuropathy, retinopathy, and nephropathy—was already recognized. However, the precise link between these complications and glycemic control had not received sufficient attention. Belgian physician Jean Pirart published a 30-year longitudinal study in 1977 (first in French, translated into English in 1978) analyzing glycemic control and complication status in 4,398 diabetic patients. Over the first 25 years of the study, nearly 21,000 examinations were conducted. The number of patients and the duration of follow-up were unprecedented at the time. Pirart and his colleagues' findings revealed the association between glycemic control and complications, and demonstrated the potential to reduce complication risk through better blood sugar management.

▋1979: The Birth of Synthetic Human Insulin
After insulin was discovered in 1921, the use of pork or beef insulin for glycemic control became widespread and saved many lives. However, insulin extracted from animal pancreases had several limitations—its blood sugar-lowering effects were variable, and it could cause allergic reactions due to the patient's immune system producing antibodies against the insulin.
With the development of recombinant DNA technology, in 1979, a research team at Genentech published a paper in PNAS reporting the first successful synthesis of human insulin. In subsequent clinical trials, synthetic human insulin not only demonstrated glucose-lowering efficacy comparable to animal-derived insulin but also showed a favorable safety profile.

▲The Genentech research team in the laboratory (Image source: Reference [2])
The U.S. FDA approved synthetic human insulin in 1982. It was the first product manufactured using genetic engineering to be approved by the FDA. This was not only a major advance in diabetes treatment but also a breakthrough in biotechnology, laying the groundwork for FDA approval of more therapeutic recombinant proteins. Today, the FDA has approved over 100 recombinant protein therapies.
▋1987: Uncovering the Role of Incretins
The concept that the gut secretes factors that stimulate insulin release after nutrient intake emerged in the early 20th century. In 1932, Belgian physiologist Jean La Barre coined the term "incretin." In the 1960s, scientists discovered that oral glucose administration stimulated greater insulin secretion than intravenous glucose, sparking a search for incretins.
In 1987, two key papers confirmed the relationship between glucagon-like peptide (GLP) and insulin secretion. Using synthetic and animal-derived GLP-1 fragments, researchers demonstrated in vitro that these peptides could stimulate insulin secretion. These studies laid the foundation for further exploration of GLP-1's role in glucose homeostasis.
We now know that GLP-1 has multiple functions, including inhibiting pancreatic glucagon secretion, slowing gastric emptying and food intake, and stimulating β cell proliferation.
Research on incretins has spurred the development of various diabetes therapies based on elevating GLP-1 levels or mimicking GLP-1 function. Today, several incretin-based therapies are widely used to treat patients with type 2 diabetes.
▋1997: The Da Qing Study Reveals the Impact of Healthy Lifestyle on Diabetes Prevention
Obesity is a major risk factor for type 2 diabetes. Over the past half-century, the explosion in type 2 diabetes incidence has paralleled the obesity epidemic. While innovative therapies have shown significant efficacy in managing type 2 diabetes, one of the most important strategies for reducing the global disease burden is preventing diabetes from developing in the first place.

In 1986, the first large-scale randomized controlled trial aimed at reducing the incidence of type 2 diabetes through behavioral interventions (including healthy diet and/or exercise) was launched in Da Qing, China. The study enrolled 577 individuals with impaired glucose tolerance, who were randomly assigned to a control group (n=138) or one of three lifestyle intervention groups (n=438): diet, exercise, or diet plus exercise. The intervention period lasted 6 years.
After 6 years, the cumulative incidence of type 2 diabetes was 67.7% in the control group, 43.8% in the diet group, 41.1% in the exercise group, and 46% in the diet-plus-exercise group. This study demonstrated that a healthy diet and/or exercise could reduce the risk of developing type 2 diabetes.
In 2019, the 30-year follow-up results of the Da Qing Diabetes Prevention Study were published in The Lancet Diabetes & Endocrinology. The study showed that compared with the control group, the combined intervention group experienced a median delay in diabetes onset of 3.96 years and a 39% reduction in diabetes risk (HR 0.61, 95% CI 0.45–0.83; p=0.0015).
▋2006: Advancing Stem Cell Therapies for Diabetes
The establishment of the first human embryonic stem cell line in 1998 opened the door to using stem cell therapies for a variety of diseases. Type 1 diabetes is particularly well-suited for this strategy, as transplanting insulin-producing pancreatic β cells into patients could provide a long-term therapeutic effect, and even the possibility of a cure.
In 2006, a research team from Novocell (now Viacyte) published a paper in Nature Biotechnology describing the differentiation of human embryonic stem cells into endocrine pancreatic cells capable of secreting multiple hormones, including insulin and glucagon.

In 2008, the same team demonstrated that transplanting pancreatic endoderm derived from human embryonic stem cells into mice led to the differentiation of glucose-responsive endocrine cells in vivo, and showed that these cells could help protect the mice from hyperglycemia.
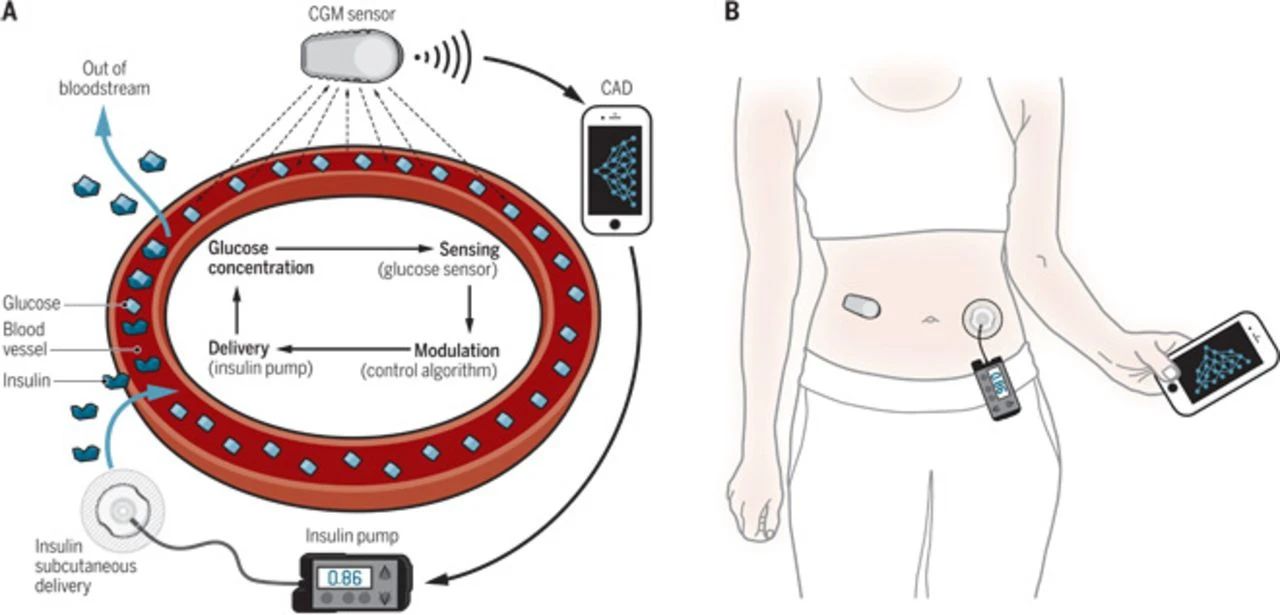
▋2014: "Artificial Pancreas" Systems Transform Patients' Lives
For patients with type 1 diabetes, maintaining blood glucose levels within the normal range can delay or prevent complications. However, the burden of having to monitor blood glucose multiple times a day and calculate insulin doses accordingly is significant.
Advances in continuous glucose monitoring systems and insulin delivery devices have finally enabled the development of "artificial pancreas" systems that combine glucose monitoring with insulin delivery. These systems use algorithms to calculate insulin doses and automatically administer the drug, greatly reducing the burden on patients.

Another meta-analysis published in 2019 found that GLP-1 receptor agonists also reduced MACE incidence by 12% across multiple clinical trials.

These additional benefits of next-generation glucose-lowering therapies have revolutionized the management of type 2 diabetes, providing patients and physicians with more options and reducing the burden of chronic complications associated with diabetes.
References:
[1] Milestones in diabetes. Retrieved June 17, 2021, from https://www.nature.com/immersive/d42859-021-00002-5/index.html
[2] CLONING INSULIN. Retrieved June 17, 2021, from https://www.gene.com/stories/cloning-insulin
[3] Boughton and Hovorka, (2019). Advances in artificial pancreas systems. Science Translational Medicine, DOI: 10.1126/scitranslmed.aaw4949