



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2021-05-14
Posting Date:2021-05-14 Views:
Views: Today, The Lancet published important data from a large randomized trial in the UK. By evaluating combination regimens of the AstraZeneca COVID-19 vaccine ChAdOx1 nCoV-19 and the Pfizer/BioNTech COVID-19 vaccine BNT162b2, the study provides preliminary safety data on mix-and-match vaccination, analyzing local and systemic reactions in participants, as well as laboratory blood and biochemical test results.

The Com-COV study, led by a team from the University of Oxford, is a multicenter, participant-blinded, randomized heterologous prime-boost COVID-19 vaccination study in the UK. It aims to provide evidence to guide different vaccine combination strategies, in order to offer greater flexibility in vaccination schedules and facilitate timely vaccination for more people.
A total of 463 participants had a two-dose interval of 28 days; all received the first dose, and 461 received the second booster dose. The median age of the 463 participants was 57 years (range 50-69), 46% were female, 25% were from ethnic minority groups, and baseline characteristics were balanced across the four different "mix-and-match" groups.

Among recipients of the ChAd/BNT combination, 34% (37 of 110) reported this reaction, compared with 10% (11 of 112) among recipients of two doses of ChAd, an absolute difference of 24% (95% CI 13%-35%).
Among recipients of the BNT/ChAd combination, 41% (47 of 114) reported this reaction, compared with 21% (24 of 112) among recipients of two doses of BNT, an absolute difference of 21% (95% CI 8%-33%).
Similar trends were observed for chills, fatigue, headache, joint pain, malaise, and muscle soreness. No participants were hospitalized due to these symptoms. Most reactions occurred within 48 hours after vaccination.
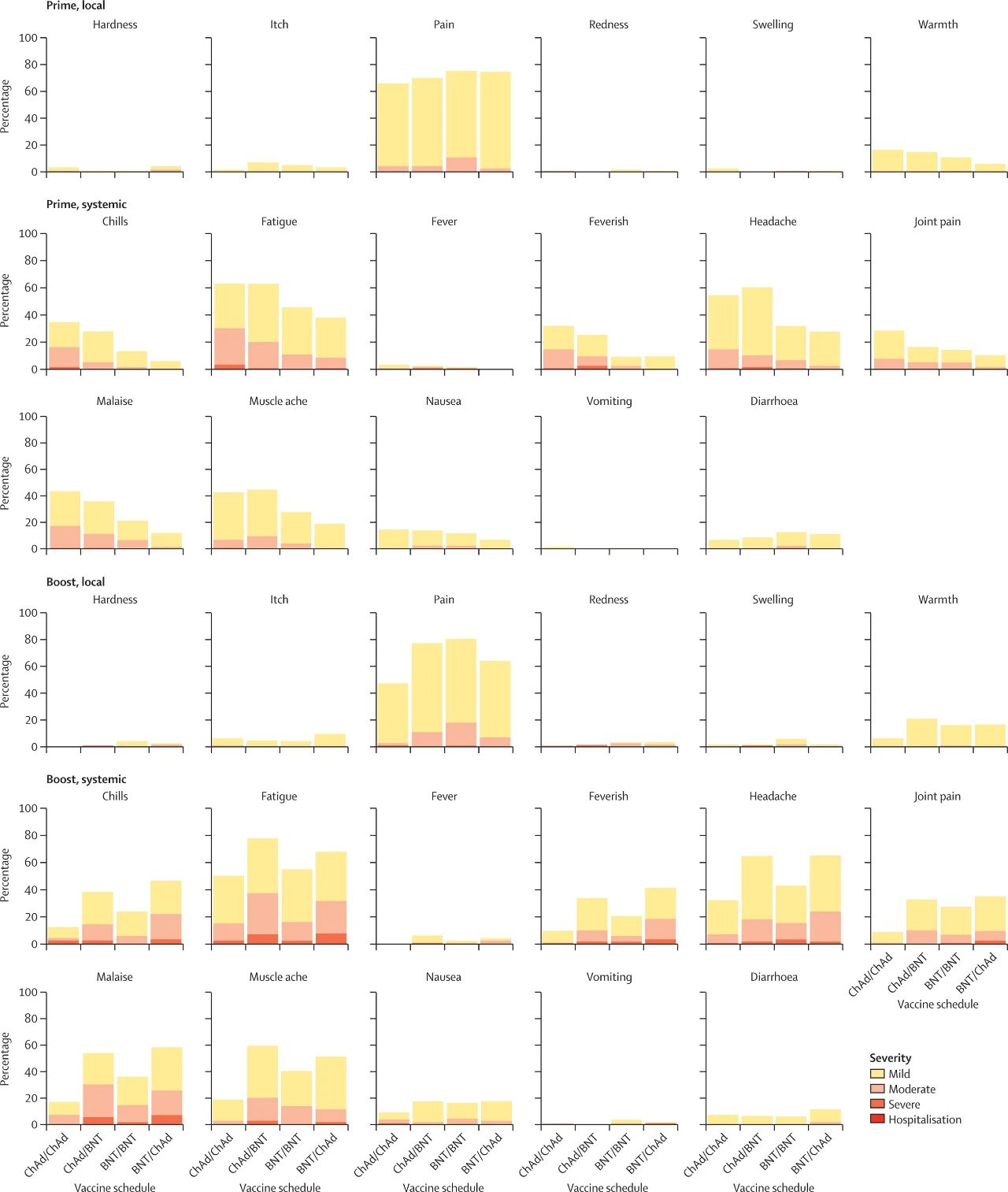
▲Self-reported local and systemic reactions, and their severity, within 7 days after the first dose (prime, top three rows) and within 7 days after the second dose (boost, bottom three rows). (Image source: Reference [1]; click to enlarge)
Local reactions include: hardening, itching, pain, redness, swelling, and warmth at the injection site.Systemic reactions include: chills, fatigue, fever (≥38.0°C), feverishness (without temperature recorded), headache, joint pain, malaise, muscle soreness, nausea, vomiting, and diarrhea.
Severity levels: mild, moderate, severe, and hospitalization.
Two doses of ChAd: 36% (40 of 112),
ChAd/BNT combination: 57% (63 of 110),
Two doses of BNT: 41% (48 of 117),
BNT/ChAd combination: 60% (68 of 114).
Hematologic and biochemical profiles were similar between participants receiving two doses of the same vaccine and those receiving mix-and-match regimens. All laboratory adverse events in the mix-and-match groups were grade 2 or lower; no thrombocytopenia was observed in any group on day 7 after the booster dose.
Dr. Matthew Snape, Professor of Vaccinology at the University of Oxford and corresponding author of the study, added, "Importantly, there are no safety issues or signals, but this does not tell us whether the immune response will be affected. We hope to report these data in the coming months."
The Com-COV study is ongoing, evaluating the long-term effects of heterologous BNT162b2 and ChAdOx1 nCoV-19 vaccination regimens. Primary immunological outcome data are expected in June 2021, and the study will also include additional analyses on whether prophylactic or routine use of acetaminophen reduces these reactions. Furthermore, the research team will evaluate combination regimens of other available COVID-19 vaccines. Longer-term data are expected to provide further insights into mix-and-match vaccination.