



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2021-04-28
Posting Date:2021-04-28 Views:
Views: Elevated serum urate concentration is the most important risk factor for the development of gout, and long-term reduction of urate levels to reverse hyperuricemia is the main strategy for effective gout management. How to manage acute gout flares, what are the key considerations for long-term management, and how can recurrence be prevented? A recent review article published in The Lancet provides a comprehensive overview of the latest advances in the clinical features, pathophysiology, and treatment of gout.

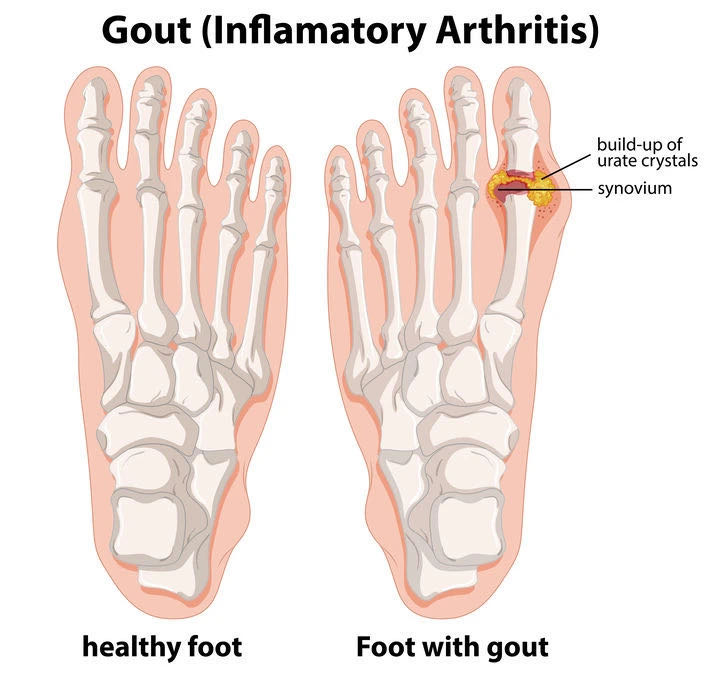
▉Four Pathophysiological Stages: Hyperuricemia ≠ Gout
The progression of hyperuricemia and gout involves four pathophysiological stages: the development of hyperuricemia, the deposition of urate crystals, gout flares triggered by acute inflammatory responses to deposited crystals, and the advanced stage characterized by tophi. Some patients may progress to the advanced stage without ever experiencing a gout flare.
Tophi represent the body's chronic, foreign-body granulomatous inflammatory response to urate crystals.
▉Clinical Manifestations Go Beyond Gout Flares
The classic initial presentation of gout is a gout flare—an acutely painful inflammatory arthritis that primarily affects the joints of the lower extremities (feet, ankles, and knees). Flares can also occur in the elbows, wrists, and hands, but upper extremity involvement is typically seen only in patients with long-standing, poorly controlled disease. Gout flares are usually monoarticular; polyarticular flares typically occur in patients with poorly controlled disease and may be accompanied by significant systemic symptoms, including fever, chills, and even delirium.
▲Gout flares primarily affect joints of the lower extremities
Gout is also often associated with multiple comorbidities, including hypertension, obesity, cardiovascular disease, diabetes, dyslipidemia, chronic kidney disease, and kidney stones. These comorbidities can complicate gout management and increase the risk of premature death.
▉Acute Gout Flares: Early Anti-inflammatory Treatment
For asymptomatic hyperuricemia, preventive measures such as dietary modification and weight loss should be considered. Urate-lowering drugs or anti-inflammatory agents are not currently recommended for gout prevention in this population.
NSAIDs should be avoided in patients with kidney disease, heart disease, peptic ulcer disease, or those taking anticoagulants.
High-dose oral prednisone should be avoided in the setting of infection, fluid retention, or diabetes.
Colchicine can be toxic in patients with severe kidney disease, severe liver disease, or those concurrently using ABCB1 inhibitors or CYP3A4 inhibitors (e.g., cyclosporine, ketoconazole, clarithromycin, and verapamil).
Additionally, clinical trials have shown:
Oral prednisolone, NSAIDs (including non-selective or COX-2 selective drugs), and low-dose colchicine have comparable efficacy for acute gout flares.
However, oral corticosteroids may have a better safety profile than NSAIDs, and NSAIDs have fewer side effects than low-dose colchicine.
Low-dose colchicine (1.0–1.2 mg immediately, followed by 0.5–0.6 mg one hour later) has comparable efficacy to high-dose colchicine (4.8 mg over 6 hours) but with fewer side effects. High-dose colchicine is not recommended.
At the same time, all patients experiencing a gout flare need to be informed about long-term effective urate-lowering therapy to prevent recurrence and joint damage.
▉Long-term Continuous Treatment to Achieve and Maintain Target Urate Levels
The main strategy for effective long-term gout management is a treat-to-target approach for serum urate, achieving sustained urate reduction to dissolve urate crystals. Clinical trials have demonstrated that this treatment strategy reduces gout flares, resolves tophi, and prevents joint damage in the long term.
For most patients with gout, the target serum urate level should be below 360 μmol/L. For those with a high urate burden (e.g., those with existing tophi), a more stringent target of ≤300 μmol/L is recommended.

When is urate-lowering therapy indicated?
Urate-lowering therapy is generally recommended for patients with:
Tophi, imaging evidence of joint damage due to gout, frequent gout flares (≥2 per year), or urolithiasis;
Younger patients (<40 years) with very high serum urate concentrations (>480 μmol/L) or comorbidities (kidney or heart disease).
During this period, low-dose anti-inflammatory drugs can be used to prevent gout flares (e.g., naproxen 250 mg daily, or colchicine 0.6 mg daily, for 3–6 months).
Choice of Urate-Lowering Drugs
Additionally, uricosuric agents such as probenecid and benzbromarone can promote urate excretion and may be used as monotherapy or in combination with allopurinol. However, a potential side effect is the risk of urolithiasis. Pegloticase metabolizes urate and has been shown in clinical trials to improve gout flares and tophi.
▉Long-term Gout Management: Addressing Comorbidities and Diet
The DASH (Dietary Approaches to Stop Hypertension) diet and weight loss can help achieve modest reductions in serum urate levels.
DASH Diet: Emphasizes low fat, low salt, and low sugar, providing balanced nutrition. It recommends lean meats, poultry, and fish as primary protein sources, along with balanced intake of fruits, vegetables, dairy, whole grains, legumes, nuts, and limited fats and sweets; it also reduces salt and saturated fat intake, such as consuming low-fat milk and avoiding fatty meats.
Patients may benefit from identifying specific foods that trigger their gout flares and avoiding those dietary triggers. However, most patients cannot achieve target serum urate levels through dietary management alone.
▉Summary
Gout is common but treatable. Long-term urate lowering, particularly with medications such as allopurinol, can prevent gout flares and improve patients' quality of life. Through appropriate initiation and persistence of urate-lowering therapy, high-quality gout management can help improve patient outcomes.