Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-08-25
Posting Date:2023-08-25 Views:
Views: Since the release of the “Key Tasks for Deepening the Healthcare System Reform in the Second Half of 2023,” which set the goal of “bringing the total number of drugs procured through national and provincial centralized procurement to 450 by the end of this year,” the progress of the ninth round of centralized procurement has drawn significant industry attention.
Today, industry sources circulated a notice titled “Notice on Organizing Medical Institutions to Report Demand Quantities for the Ninth Round of National Centralized Drug Procurement” (hereinafter referred to as the “Notice”). According to the work plan in this notice, the ninth round of national centralized procurement is about to begin volume reporting. The list of varieties included in the reporting covers 44 drugs, totaling 195 specifications. (See the list at the end of the article for details.)


It is reported that the volume reporting for this round will officially begin in late August. After the hospital reporting phase concludes, the data will be reviewed by provincial medical insurance bureaus and submitted to the National Joint Procurement Office. Market analysts noted that injectable formulations account for over 60% of the varieties in this round, including several well-known blockbuster drugs. “National centralized procurement has entered a new phase of normalization and institutionalization. Policies are continuously being refined in terms of product pricing and supply security. As the reform deepens, the market is closely watching how the impact on end-users will evolve.”
“Blockbuster Drugs” Begin Volume Reporting No Biosimilars Included?
According to the “Notice,” the ninth round of national centralized procurement is expected to include 195 specifications, featuring numerous blockbuster drugs, such as dexrazoxane for injection (250mg, 500mg), lenalidomide oral immediate-release formulations (5mg, 10mg, 15mg, 25mg), rabeprazole oral immediate-release formulations (10mg, 20mg), calcium gluconate injection, atropine injection, propofol injection, oxytocin injection, levonorgestrel tablets, and sildenafil orally disintegrating tablets.
In addition, based on the selection criteria, the drugs included in this round generally meet a competitive landscape of at least “4 generics + 1 original,” with the majority being products where five or more generics have passed the consistency evaluation. A total of 44 major drugs are included (accounting for different dosage forms of three products).
Experts pointed out that based on the information disclosed in the “Notice,” the number of products included in this round is relatively modest, with injectable formulations accounting for approximately 60%. This is primarily because a large number of injectable blockbuster products have passed the consistency evaluation. Compared to previous rounds (fifth through eighth), the proportion of anti-infective injectable formulations has decreased, mainly because such drugs have largely been “procured as much as possible” in prior procurement rounds.
In fact, across the previous eight rounds of centralized procurement, over 330 drugs have been included: 25 in the first round, 32 in the second, 55 in the third, 45 in the fourth, 61 in the fifth, 16 in the sixth, 60 in the seventh, and 39 in the eighth.
Industry insiders noted that with the addition of the 44 drugs from the ninth round, the total number of drugs procured nationally will approach 380. Combined with provincial-level procurement varieties, reaching the target of 450 drugs nationally by the end of the year should be achievable.
In terms of therapeutic areas, drugs for major diseases such as antibiotics, hypertension, diabetes, and gastrointestinal conditions are heavily represented. Looking at dosage forms in this round, beyond conventional immediate-release, sustained-release, and injectable forms, the list also includes dry suspensions, chewable tablets, oral solutions, enteric-coated sustained-release tablets, orally disintegrating tablets, nebulized inhalation solutions, granules, and eye drops.
Additionally, biosimilars—particularly those with multiple approved products, such as bevacizumab injection, adalimumab injection, rituximab injection, infliximab for injection, trastuzumab for injection, and denosumab injection—have seen multiple manufacturers approved for marketing. Given that the National Healthcare Security Administration has previously signaled its intention to “include biosimilars in centralized volume-based procurement,” the industry has been closely watching for developments.
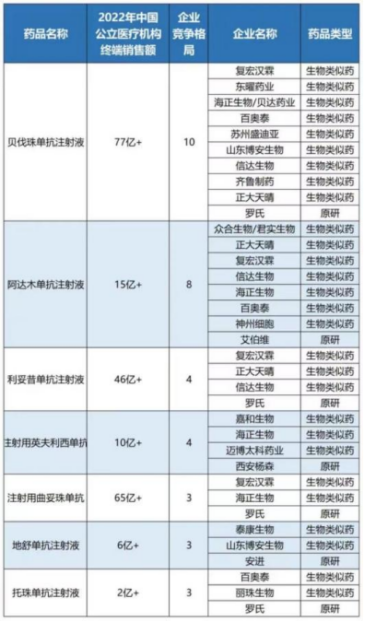
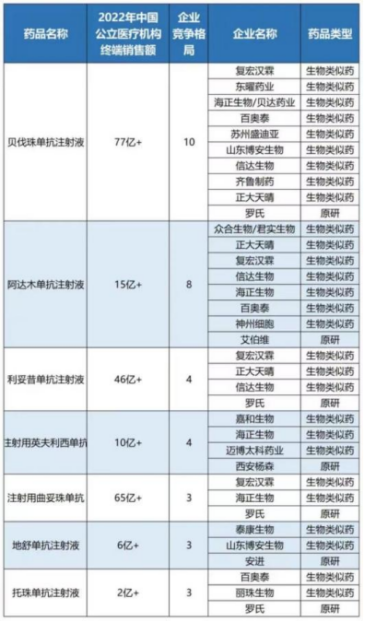
However, the aforementioned popular biosimilars did not appear in the product list disclosed in this “Notice.” Market analysts suggest that as the normalization and institutionalization of centralized drug procurement continues to advance, the inclusion of biosimilars in national procurement remains highly likely in the future.
Emphasis on “Refined” Volume Reporting “Key Monitored” Products Under Special Scrutiny
Regarding the accuracy of volume reporting, the “Notice” requires medical insurance departments at all levels to compare the reported volumes with historical procurement volumes from 2022. If a medical institution reports less than 80% of its 2022 historical procurement volume, it must provide an explanation. Institutions with historical procurement volumes that choose not to participate in reporting will be subject to heightened scrutiny.
In the volume reporting documents for the ninth round of centralized procurement, the trend toward refined management is further evident:
The “Notice” specifically noted that for drugs newly added to the national reimbursement drug list in 2021 and 2022, such as esmolol injection and citicoline injection, the reported volumes should strive to reach 100% of historical procurement volumes.
For nationally key monitored drugs, such as rabeprazole oral immediate-release formulations, esomeprazole magnesium enteric-coated dry suspension, and piracetam injection, no lower limit has been set for medical institutions’ reported volumes.
This reflects the heightened attention paid to “nationally key monitored drugs” in centralized procurement. Indeed, since January of this year, when the National Health Commission issued the “Notice on Issuing the Second Batch of Nationally Key Monitored Rational Use Drug List,” 17 provinces have followed up by updating their own key monitoring lists.
Three drugs from the second batch of key monitored drugs are included in the ninth round: rabeprazole oral immediate-release formulations, esomeprazole magnesium enteric-coated dry suspension, and piracetam injection.
Proton pump inhibitors are commonly used acid-suppressing drugs in clinical practice. Data from Minenet shows that in the first half of 2022, sales of proton pump inhibitors in China’s public medical institutions exceeded RMB 9 billion. Among the top 10 proton pump inhibitor products, injectable and oral immediate-release formulations each accounted for five. Omeprazole sodium for injection, rabeprazole sodium enteric-coated capsules, and rabeprazole sodium enteric-coated tablets ranked first, second, and third, respectively. Among these, sales of omeprazole sodium for injection and rabeprazole sodium enteric-coated capsules each exceeded RMB 1 billion in the first half of the year.
Esomeprazole magnesium enteric-coated dry suspension, developed by AstraZeneca, is primarily indicated for gastroesophageal reflux disease, treatment of reflux esophagitis, long-term prevention of relapse in patients with healed esophagitis, and symptom control of gastroesophageal reflux disease. Compared with conventional formulations, it is easier to administer to pediatric patients and those with swallowing difficulties. In 2022, sales of esomeprazole in China’s public medical institutions exceeded RMB 1 billion. Currently, four dosage forms—injection, capsule, tablet, and oral liquid—have been approved for marketing. The esomeprazole magnesium enteric-coated dry suspension has production approvals from five companies, including Zhejiang Erying Pharmaceuticals, Otsuka Pharmaceutical, and Easton Biopharmaceuticals, all of which were approved this year.
Piracetam injection, used for cardiovascular and cerebrovascular conditions, is indicated for memory impairment and mild to moderate brain dysfunction caused by acute and chronic cerebrovascular diseases, traumatic brain injury, various toxic encephalopathies, and other causes. It can also be used for delayed intellectual development in children. Piracetam injection is a Category B drug in the National Reimbursement Drug List (2020). Data shows that sales of piracetam in urban public hospitals in China have grown steadily over the past three years, reaching RMB 1.58 billion in 2021.
Market observers believe that the changes in procurement rules seen in the eighth round, compared to the seventh, may carry over into the ninth round. These include more standardized procurement cycles, the “one main, two backup” approach for some varieties, and the option for reverse selection. Factors such as price comparison ratios and backup supply product prices also warrant close attention. The ninth round of national centralized procurement is one of the major focal points for the second half of the year. Combined with the ongoing anti-corruption efforts in the pharmaceutical sector, price reductions for some products may reach historic lows.