



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-06-30
Posting Date:2023-06-30 Views:
Views:
Source: Dashan Pharmaceutical Formulation Department Editor: Zhang Tao
I. Background
During the generic development of injectable products, it is common to encounter overage where the actual amount exceeds the labeled amount. Overage includes both overfill and overage in drug substance input. This article will briefly organize and discuss the study of overfill in injectable products to help better understand the relevant concepts.
II. Overview
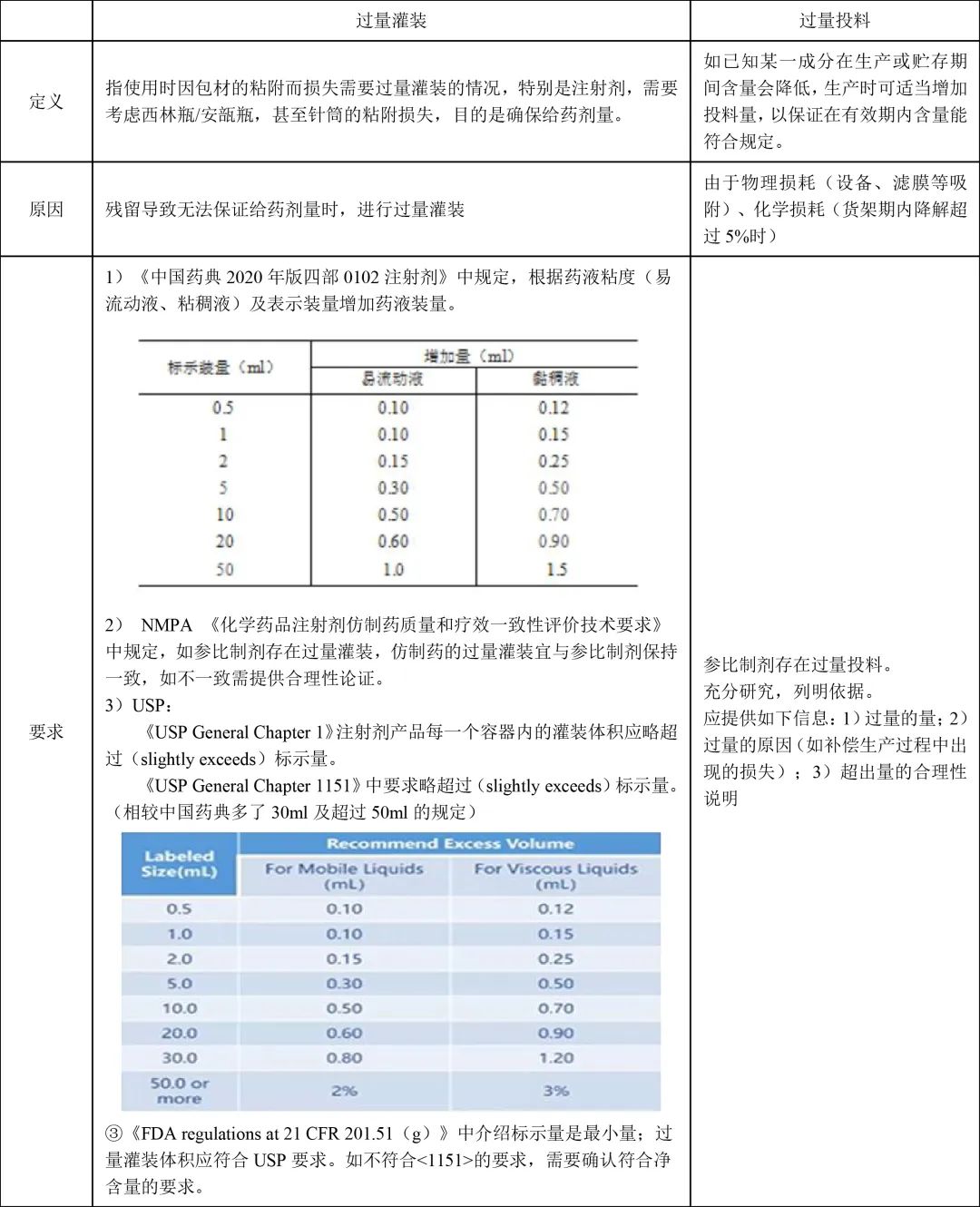
III. Study Methods
1. Key Concepts

Translation:
Withdrawable Volume: The volume of liquid drug product, or liquid constituted from solid drug product, that can be transferred from the original container according to the procedure outlined in USP General Chapter <697> Container Content for Injections.
Net Content: The amount of net contents as specified in 21 CFR 201.51(g). Net content should appear as a separate item on the label. For injectable products marketed: Liquids – container net content is expressed in units of volume (e.g., mL); Solids – container net content is expressed in units of weight (e.g., mg).
Gross Content: (1) For liquid drug products: the total volume of drug product filled into the vial, including overfill. (2) For solid drug products: the total amount of drug substance or protein content filled into the vial (e.g., mg), including overage. The study of overfill primarily revolves around these three concepts. In addition, due to clinical administration requirements, several other concepts may exist as follows:
Deliverable Volume: The volume that can be expelled from the administration device.
Residual Volume: 1) Container residual volume: the volume of drug product remaining in the container after withdrawal. 2) Administration device residual volume: the volume of drug product remaining in the administration device after expulsion.
2. Study Methods
2.1 Withdrawable Volume Measurement Methods:
1) Measurement using a graduated cylinder after withdrawal with a syringe. (Use a “to contain” graduated cylinder, with the size such that the volume to be measured is at least 40% of its nominal capacity; suitable for low-viscosity drug products; for high-viscosity drug products, refer to Chinese Pharmacopoeia General Chapter 0942.)
2) Measure the density of the drug product and the weight of the withdrawable drug product, then calculate the volume. Requirements for the syringe used:
① The syringe volume should be such that 40% of it is greater than the specification of the injectable product being tested.
② USP uses gauge 21; domestically, gauge 7 or 8 needles are used.
2.2 Overfill Study for Generic Products
1) Injections
① Literature: CMC review reports or instructions for use.
② Measure the weight and density of the drug product.
③ Measure the withdrawable volume. Determine the weight of the reference product “drug product + empty vial” (A). Determine the weight of “residual drug product + empty vial” after withdrawal (B). Determine the weight of the “empty vial” after drying (C). Withdrawable volume (D): D = (A - B) / ρ. Total volume of drug product (gross content) (E): E = (A - C) / ρ. Reference product overfill calculation: E / labeled volume (mL). Reference product residual volume (F): F = E - D. Self-developed product fill volume: reference withdrawable volume (D) + self-developed product residual volume (F’).
④ Determine the drug content per container: Determine the drug content per container (A) (mg): After rinsing and fully transferring the contents of one container, dilute and inject for analysis to calculate the drug content per container (mg). Reference product overfill calculation: drug content per container (A) / labeled amount. Self-developed product fill volume: can be determined accordingly.
⑤ Mark the liquid level, add water to the mark, weigh the added water, calculate the volume through density conversion, and determine the fill volume.
2) Lyophilized Products
(1) Lyophilized Products:
① Literature: CMC review reports or instructions for use.
② Determine the weight and moisture content of the contents. For sterile powders, the sample size for the uniformity of dosage units test for injections should be used.
③ Determine the API content of the entire container (has limitations; not recommended for products with poor stability).
④ Determine the excipient content (e.g., mannitol; stable excipients).
⑤ Mark the liquid level, add water to the mark, weigh the added water (less accurate).
⑥ Not following clinical practice: Determine the weight of the reference product “drug product + empty vial” (A). Determine the weight of the “empty vial” after drying (C). Determine the content of API and excipients in the drug product (g/g) (content by weight; determine the API and excipient content per gram of drug product): (E). Reference product overfill calculation: (A - C) × E / labeled amount.
⑦ Following clinical practice: Reconstitute according to the clinical use volume. Determine the weight of the reference product “drug product + empty vial” (A). Determine the weight of “remaining drug product + empty vial” after withdrawal (B). Determine the weight of the “empty vial” after drying (C). Withdrawable drug product weight (D): D = A - B. Determine the content of API and excipients in the drug product (g/g) (content by weight; determine the API and excipient content per gram of drug product): (E). Residual drug product weight in the container (F): F = B - C. Reference product overfill calculation: (A - C) × E / labeled amount. Self-developed product fill volume: reference withdrawable weight (D) + self-developed product residual weight (F).
2.3 Fill Volume Control During Manufacturing
1) Regulatory Requirements for Product Quality
Small Volume Injections
Fill volume requirements in Chinese Pharmacopoeia 2020 Edition, Part IV, General Chapter 0102: The fill volume should not be less than the labeled volume.
USP <698>

Translation: The average liquid volume obtained from 10 containers is NLT 100%, and the volume in each of the 10 containers is within the range of 95%–110% of the volume declared on the label. If (A) the average volume is less than 100% of the volume declared on the label and none of the containers is outside the range of 95%–110%, or if (B) the average volume is NLT 100% and NMT 1 container is outside the range of 95%–110% but within the range of 90%–115%, then test an additional 20 containers. The average liquid volume obtained from these 30 containers is NLT 100% of the volume declared on the label; the volume from NMT 1 of the 30 containers is outside the range of 95%–110% but within the range of 90%–115% of the volume declared on the label.
Lyophilized Products
Fill volume requirements in Chinese Pharmacopoeia 2020 Edition, Part IV, General Chapter 0102: The uniformity of dosage units requirements are as follows:

2) Principles
The theoretical lower limit of the drug product fill volume should meet the pharmacopoeial fill volume requirements. The target fill volume can be determined with reference to the overfill volume of the reference product. Combining these with the error range of the filling equipment, the fill volume control during the manufacturing process can be established.
3) Considerations
Pay attention to potential differences between the devices or instruments used for fill volume measurement during line adjustments in the manufacturing facility and those used in laboratory studies. Calibration to eliminate these discrepancies should be performed to determine the appropriate fill volume.