



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-06-16
Posting Date:2023-06-16 Views:
Views: Under the impact of centralized procurement and the pandemic, the market for chemical drugs for respiratory system in China’s public medical institutions has experienced significant fluctuations and a reshuffling of the competitive landscape. Currently, 76 chemical drugs for respiratory system have been approved by or deemed equivalent to passing the generic drug consistency evaluation by manufacturers. Yangtze River Pharmaceutical Group and Beite Pharmaceutical lead the list in terms of the number of approved generics. 21 varieties (by generic name) have been included in national centralized procurement, with several top-selling drugs experiencing significant sales declines. Seven varieties are poised for inclusion in the ninth round of centralized procurement, with multiple varieties from companies such as Yangtze River Pharmaceutical Group, Renhe Yikang, Beite Pharmaceutical, Fu’an Pharmaceutical, Yipinhong, and Joincare listed.
Market Fluctuations! “Top-Selling Drug” Sales Halved, 2 Inhalation Drugs Surge Over 150%
Under the impact of centralized procurement and the pandemic, the market for chemical drugs for respiratory system in China’s urban public hospitals, county-level public hospitals, community health centers, and township health centers (collectively referred to as public medical institutions in China) has experienced significant volatility. In 2022, the sales scale approached RMB 40 billion (a decrease of nearly RMB 14 billion compared to the peak period in 2019), representing a year-on-year decline of 11.85%.
Recent Sales Performance of Chemical Drugs for Respiratory System in China’s Public Medical Institutions (Unit: RMB 100 million)
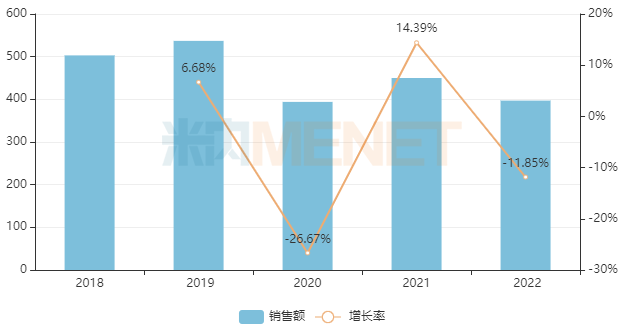
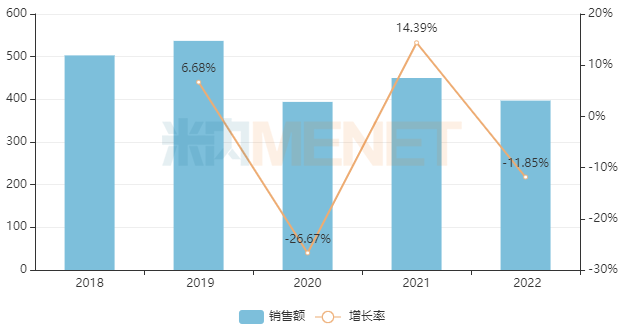
By subcategory, drugs for obstructive airway diseases dominate the chemical drugs for respiratory system market, accounting for nearly 49% of the share. This is followed by cough and cold preparations at nearly 30%, and systemic antihistamines rank third, accounting for nearly 12%. In terms of enterprise rankings, in 2022, domestic companies such as Yangtze River Guangzhou Hairui Pharmaceutical, Joincare Pharmaceutical Group, and Sichuan Purent Pharmaceutical saw significant improvements in their rankings. Notably, Joincare Pharmaceutical Group and Sichuan Purent Pharmaceutical both recorded sales growth rates exceeding 140%.
Among the top 20 chemical drug products for respiratory system, six products achieved annual sales exceeding RMB 1 billion. Budesonide Suspension for Inhalation, Budesonide Formoterol Powder for Inhalation (II), and Acetylcysteine Solution for Inhalation ranked in the top three, with sales exceeding RMB 3.1 billion, RMB 2.2 billion, and RMB 2.0 billion, respectively.
Top 20 Chemical Drug Products for Respiratory System in China’s Public Medical Institutions in 2022
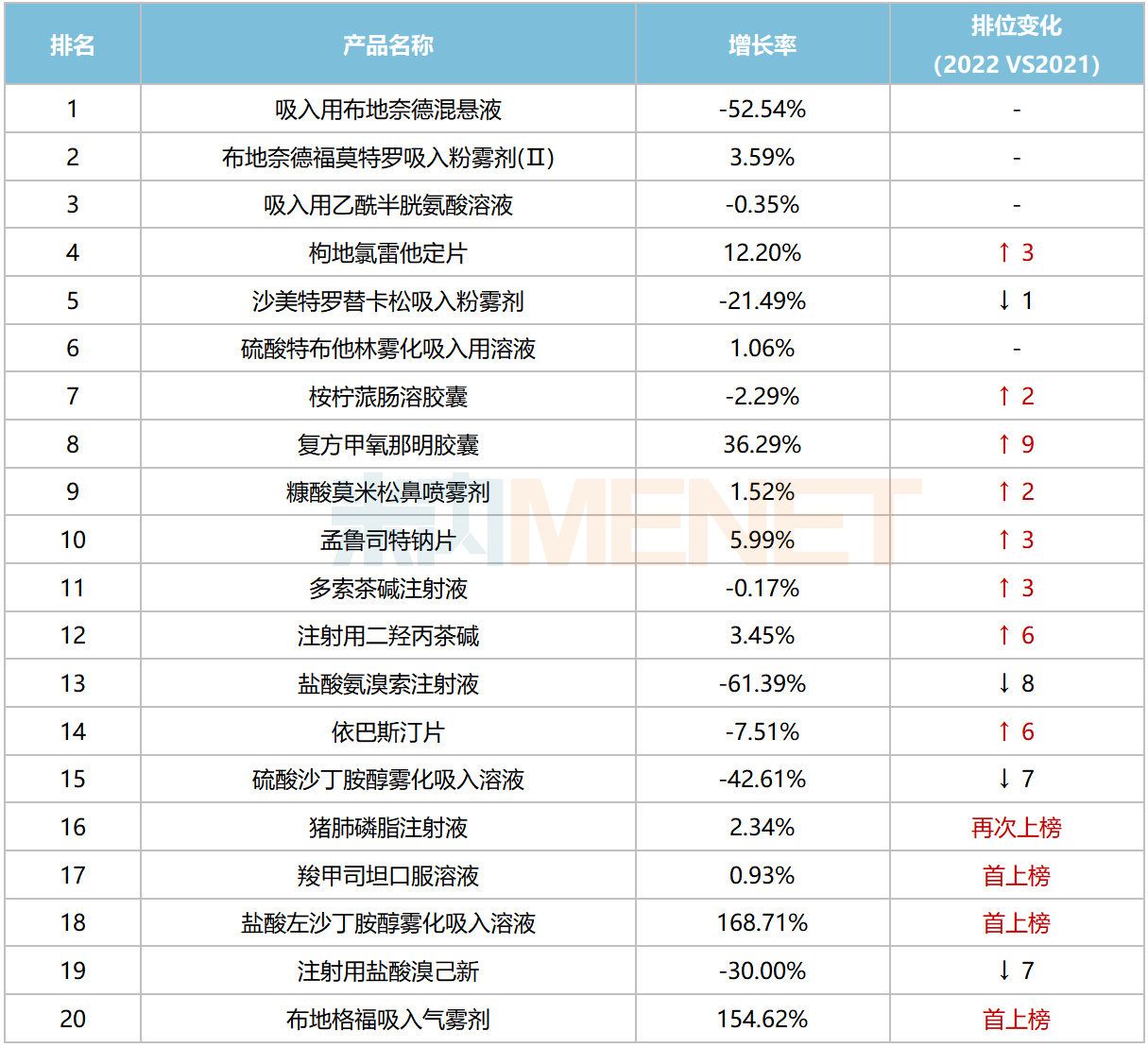
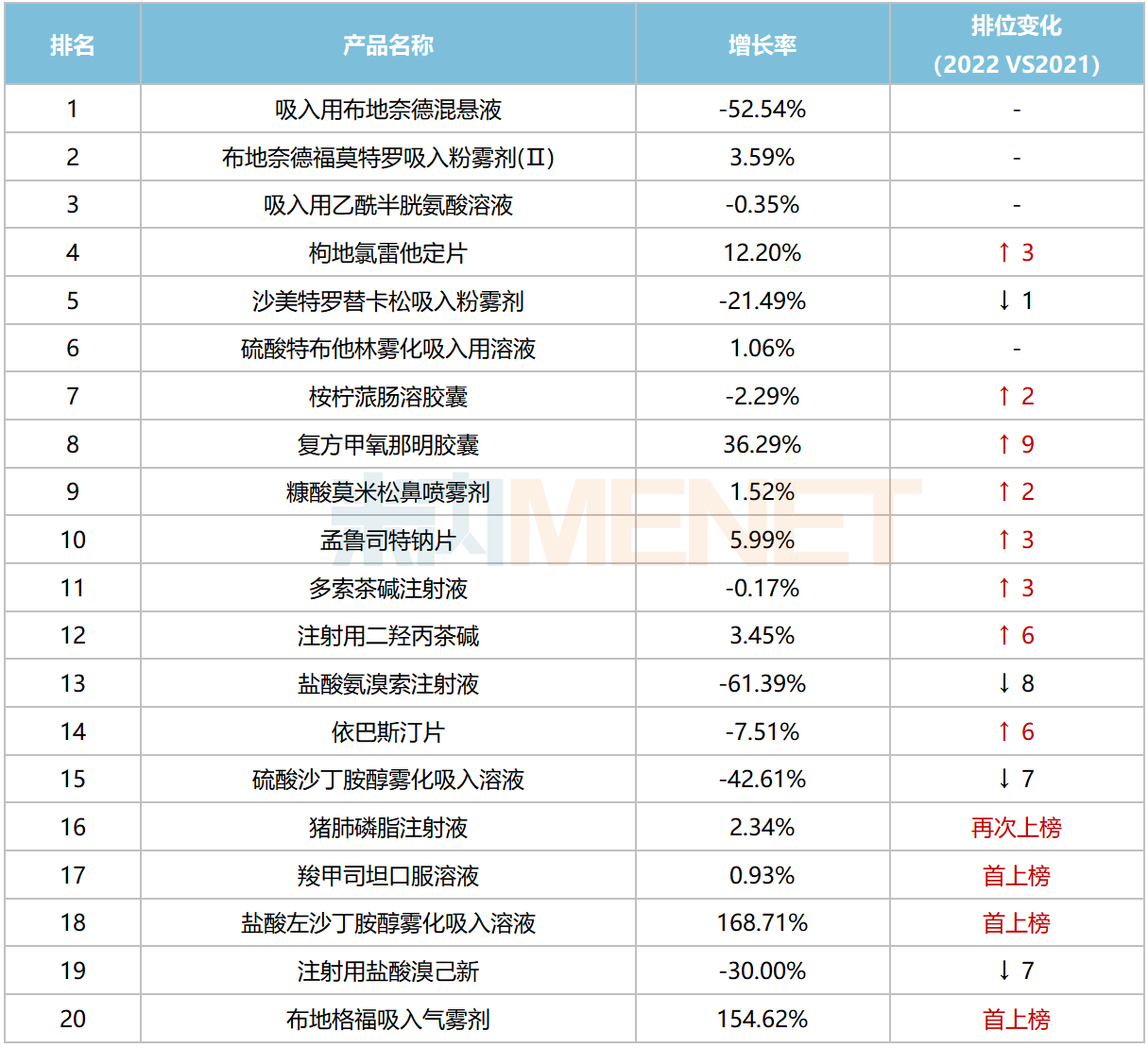
In terms of sales growth rates, four products recorded increases exceeding 10%. Notably, two inhalation drugs—Levosalbutamol Hydrochloride Nebulizer Solution and Budesonide Glycopyrronium Formoterol Pressurized Inhalation Solution—surged over 150% and entered the top 20 for the first time. Compound Methoxyphenamine Capsules grew by over 36%, climbing nine spots compared to 2021.
Due to the implementation of centralized procurement, some products in the top 20 experienced sales declines. Among the fourth round centralized procurement varieties, Ambroxol Hydrochloride Injection, Salbutamol Sulfate Nebulizer Solution, and Doxofylline Injection fell by 61.4%, 42.6%, and 0.17%, respectively. Budesonide Suspension for Inhalation, a fifth round variety, declined by 52.5%. Among the seventh round varieties, Bromhexine Hydrochloride for Injection and Ebastine Tablets fell by 30% and 7.51%, respectively.
In the brand top 20, AstraZeneca had four brands listed, with Budesonide Formoterol Powder for Inhalation (II) and Budesonide Suspension for Inhalation ranking first and second, respectively. Among domestic brands, Yangtze River’s Desloratadine Citrate Disodium Tablets ranked third. Both Chia Tai Tianqing Pharmaceutical and Joincare Pharmaceutical each had two brands listed, with Joincare’s Levosalbutamol Hydrochloride Nebulizer Solution and Budesonide Suspension for Inhalation both achieving growth rates exceeding 140%.
Yangtze River and Beite Lead in Generic Approvals! 21 Varieties Included in National Centralized Procurement
Data shows that currently, 76 chemical drugs for respiratory system (67 generic drugs) have been approved by or deemed equivalent to passing the consistency evaluation by manufacturers, involving more than 180 pharmaceutical companies (by group).
In terms of drug variety approvals, the number of companies that have passed the consistency evaluation for Ambroxol Hydrochloride Injection, Doxofylline Injection, Montelukast Sodium Chewable Tablets, Salbutamol Sulfate Solution for Inhalation, Terbutaline Sulfate Solution for Nebulization, Ipratropium Bromide Solution for Inhalation, Ambroxol Hydrochloride Oral Solution, Levocetirizine Hydrochloride Tablets, Levosalbutamol Hydrochloride Solution for Inhalation, and Desloratadine Oral Solution all reached 10 or more.
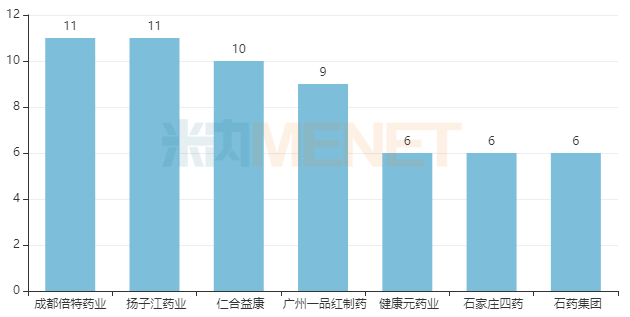
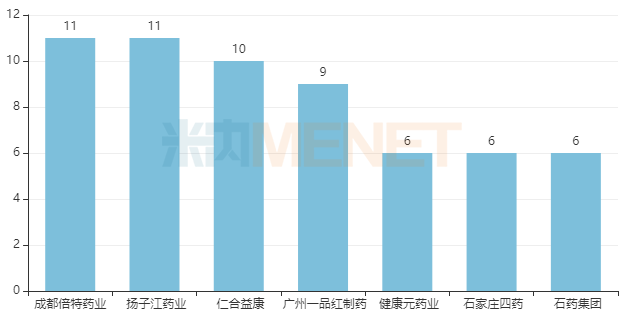
In terms of company approvals, Yangtze River Pharmaceutical Group and Chengdu Beite Pharmaceutical lead with 11 varieties each. Renhe Yikang ranks third with 10 varieties. Guangzhou Yipinhong Pharmaceutical ranks fourth with 9 varieties, while Joincare Pharmaceutical, Shijiazhuang No.4 Pharmaceutical, and CSPC Pharmaceutical are tied for fifth with 6 varieties each.
Companies with 6 or more Approved Chemical Drugs for Respiratory System
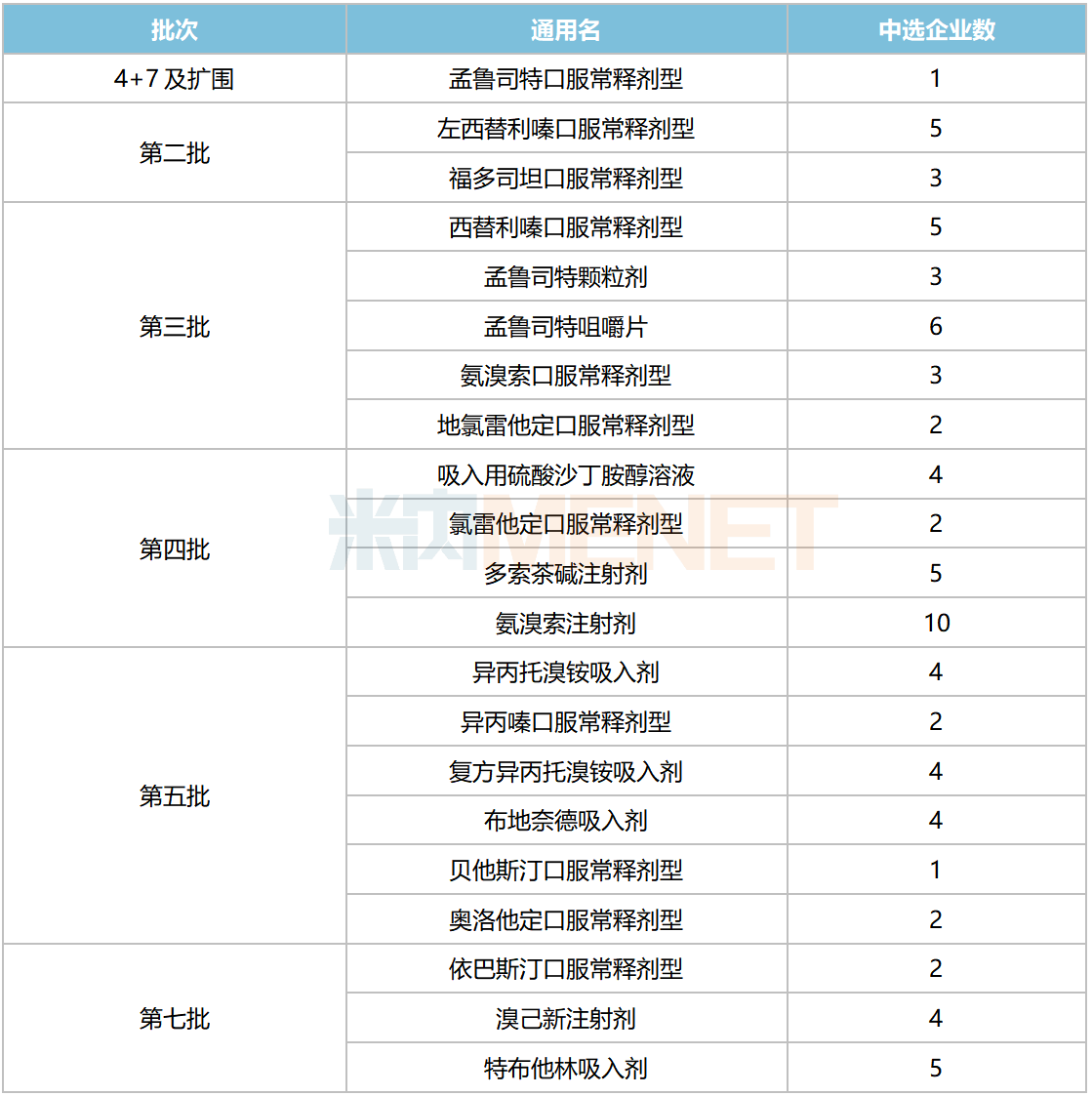
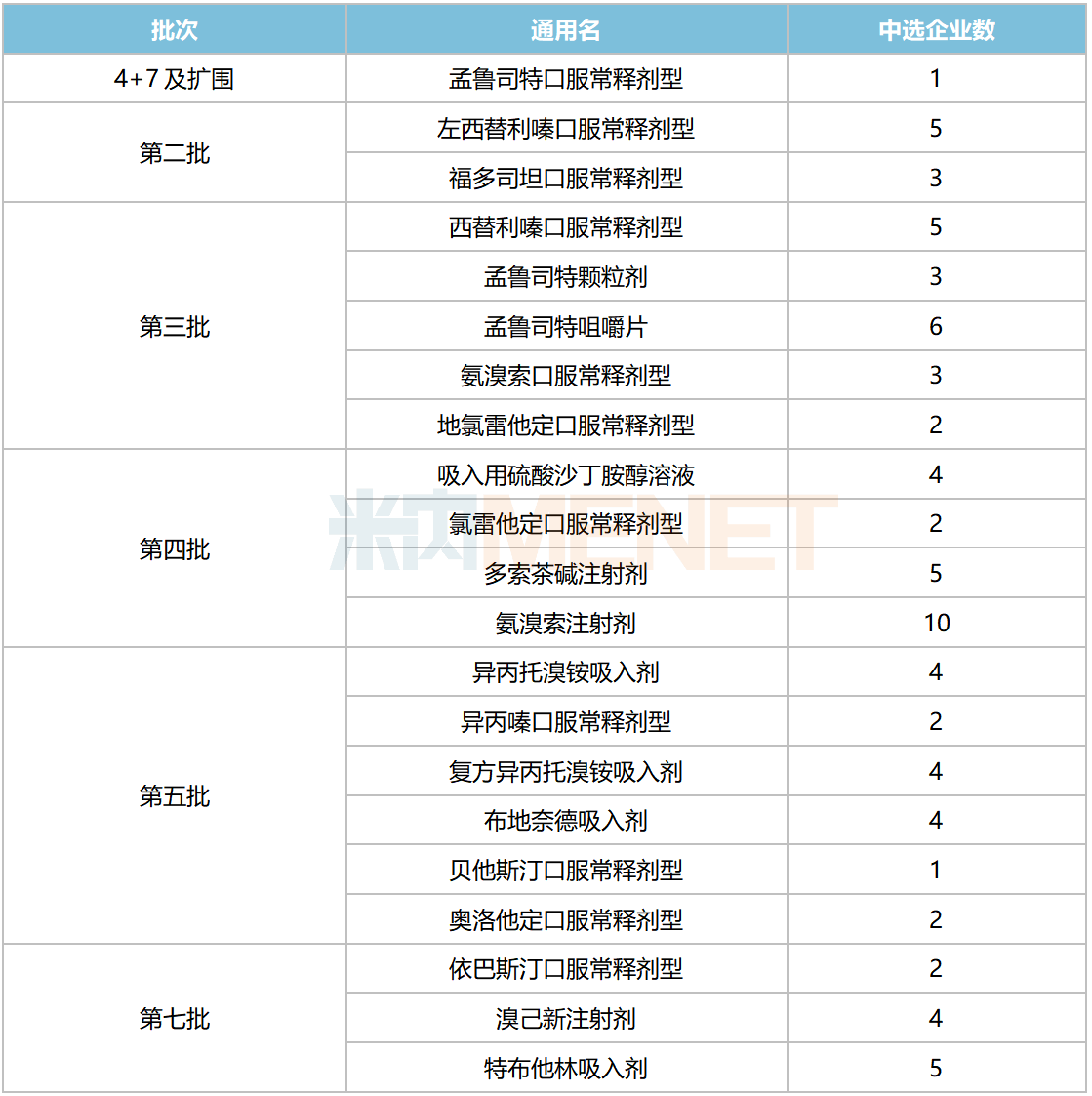
In the seven rounds (eight batches) of chemical drug centralized procurement conducted to date, respiratory system drugs have been included in quantities of 1, 2, 5, 4, 6, and 3 varieties (by generic name), totaling 21 varieties.
National Centralized Procurement for Respiratory System Chemical Drugs
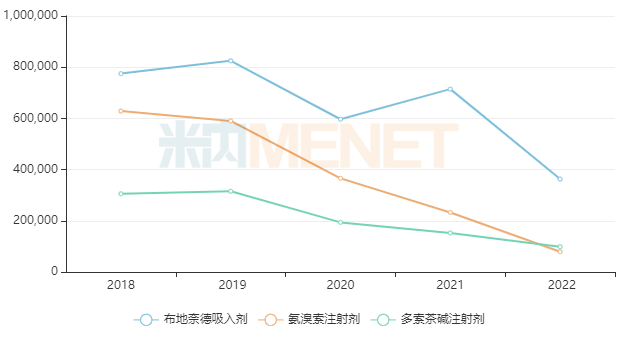
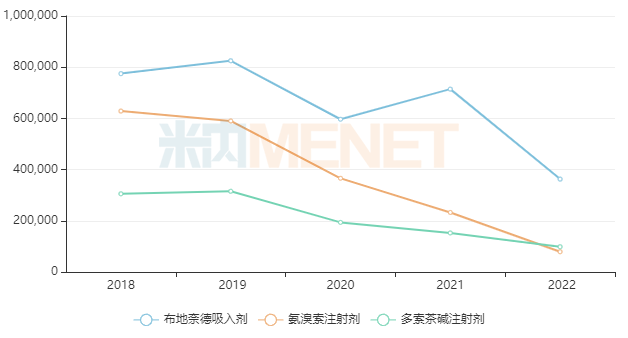
Sales of some popular existing products declined sharply after the implementation of centralized procurement. For example, Budesonide Inhalation preparations had peak sales exceeding RMB 8 billion but fell to less than RMB 4 billion in 2022. Ambroxol Injections had peak sales approaching RMB 7 billion but fell to nearly RMB 800 million in 2022. Doxofylline Injections had peak sales exceeding RMB 3 billion but fell to less than RMB 1 billion in 2022.
Sales Changes of Some Popular Varieties in China’s Public Medical Institutions After Centralized Procurement (Unit: RMB 10,000)
RMB 2 Billion Inhalation Drug in the Pipeline! 7 Varieties Poised for Ninth Round of Centralized Procurement
Excluding varieties already included in national centralized procurement, seven chemical drugs for respiratory system currently meet the procurement threshold, with the number of companies eligible to apply reaching five or more. These include four oral liquids, two inhalation drugs, and one granule formulation.
Chemical Drugs for Respiratory System with 5 or More Eligible Companies Not Yet Included in Centralized Procurement
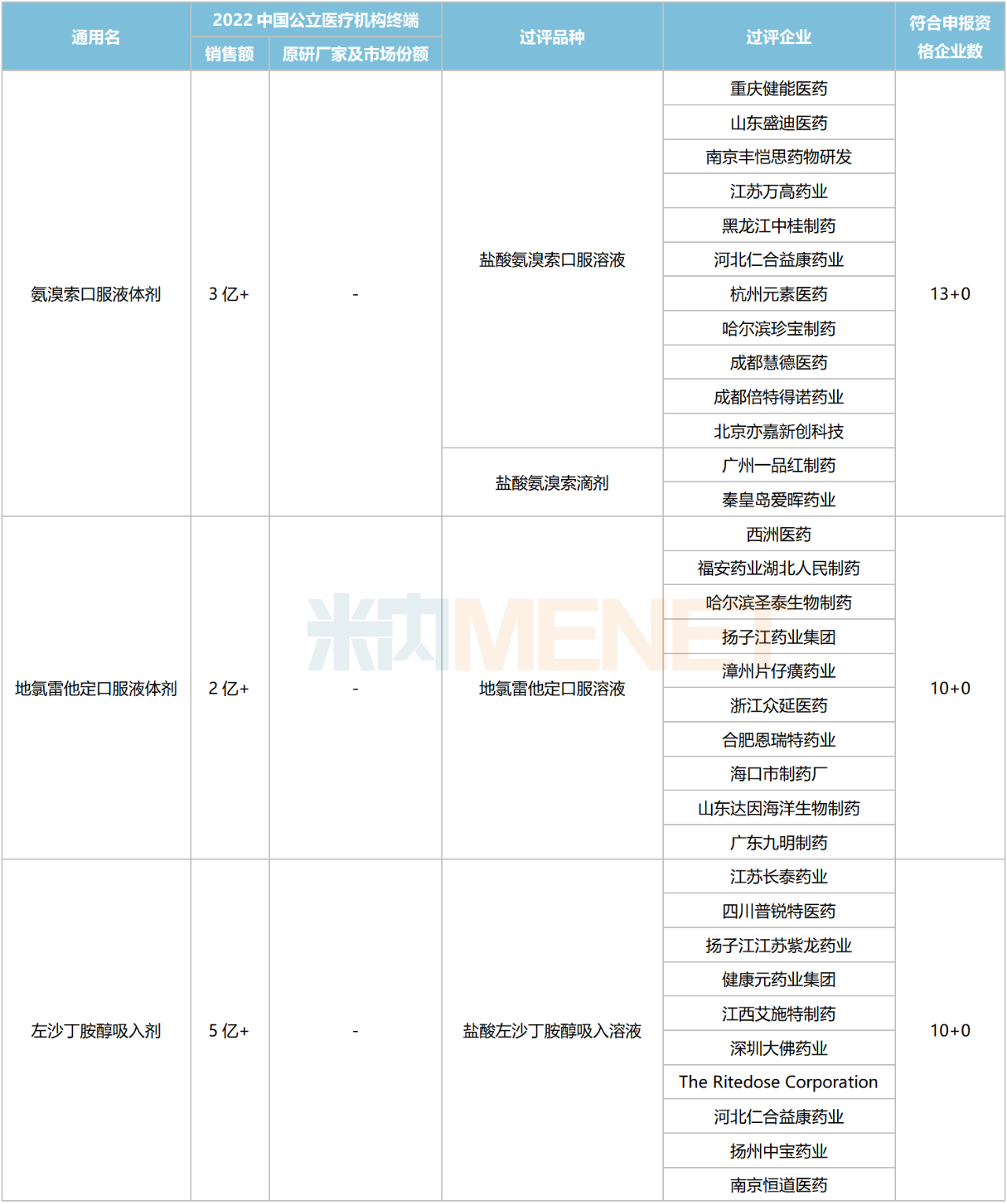
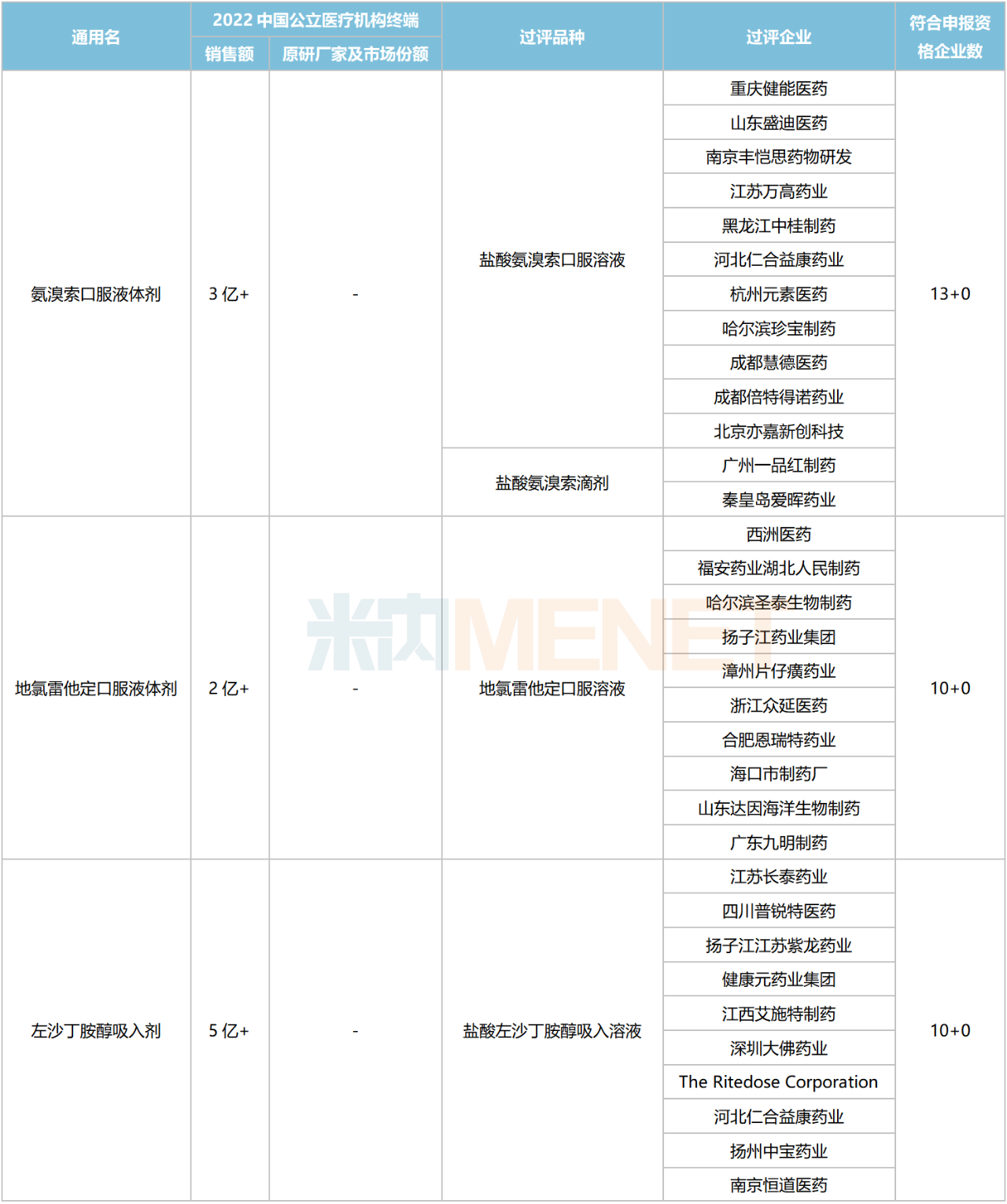
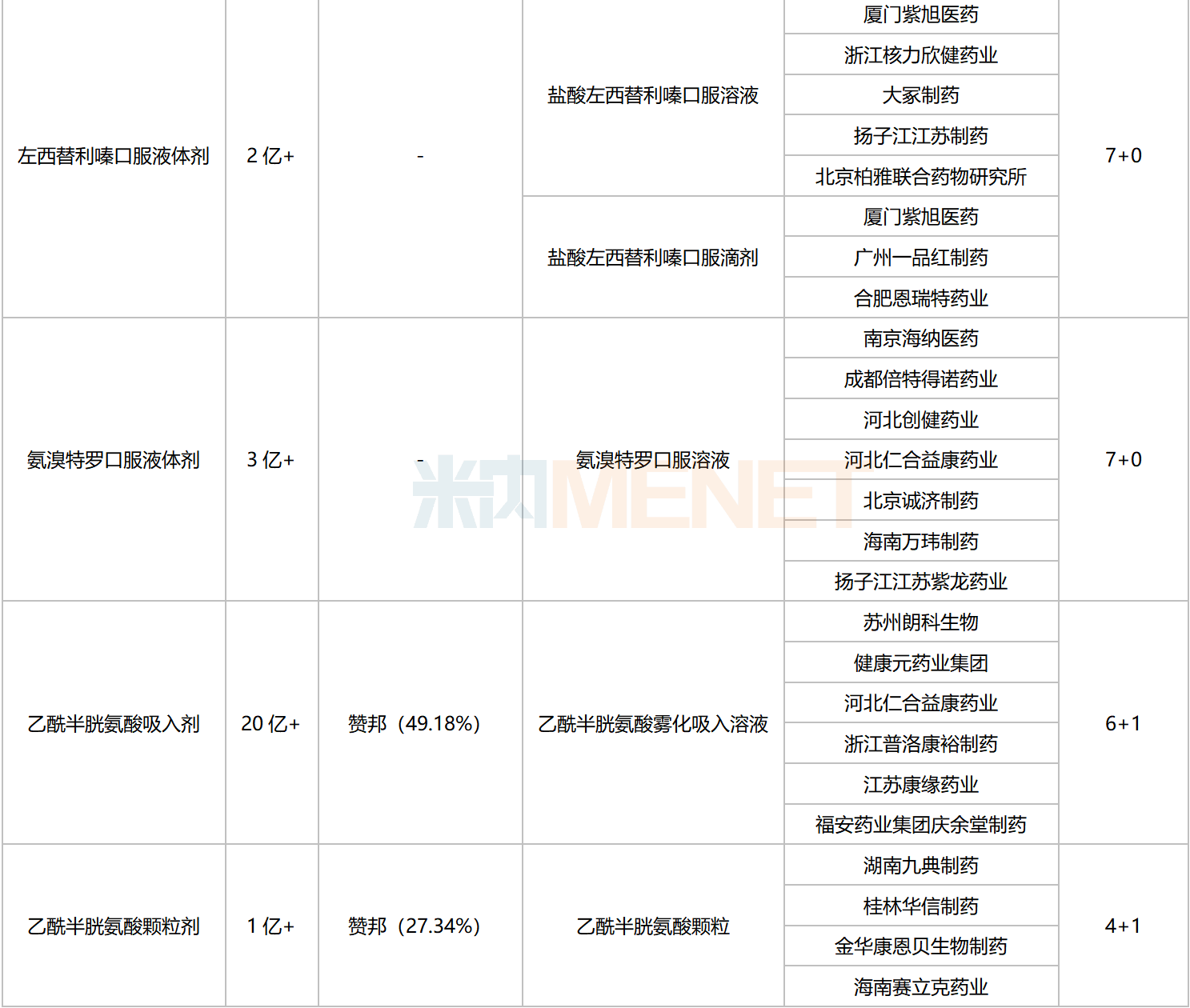
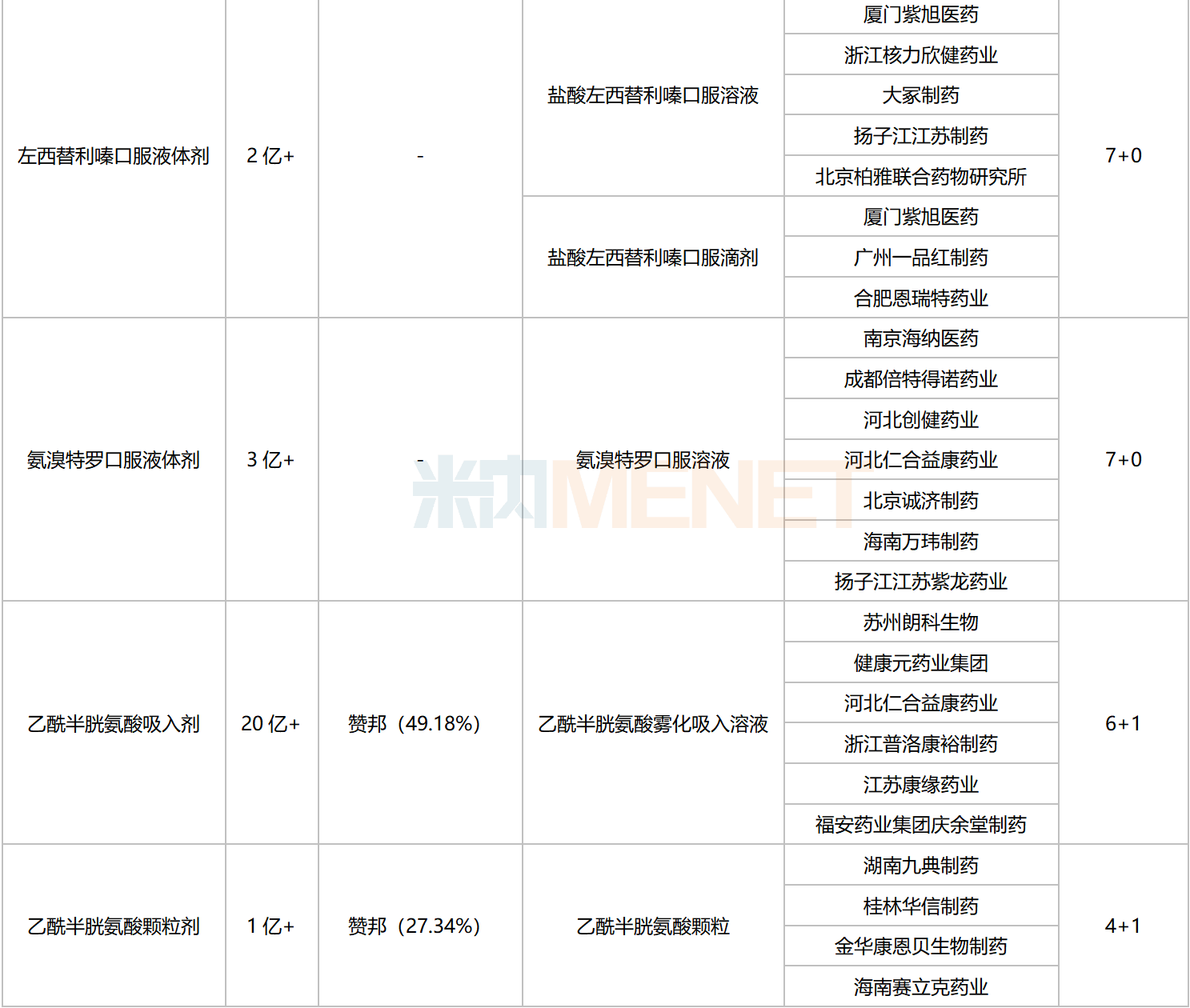
Data shows that the total sales scale of these seven varieties in China’s public medical institutions in 2022 exceeded RMB 4 billion. Acetylcysteine Inhalation preparations led with over RMB 2 billion in sales. In the ranking of chemical drug products for respiratory system in China’s public medical institutions in 2022, Acetylcysteine Solution for Inhalation ranked third.
In terms of competitive landscape, Ambroxol Oral Liquids, Desloratadine Oral Liquids, and Levosalbutamol Solution for Inhalation face intense competition, with the number of eligible companies reaching 10 or more. Acetylcysteine Granules currently have a 4+1 competitive pattern, which is relatively favorable.
Regarding companies that have passed the consistency evaluation, Yangtze River Pharmaceutical Group and Renhe Yikang each have four varieties approved. Chengdu Beite Pharmaceutical has three varieties listed, while Fu’an Pharmaceutical, Guangzhou Yipinhong Pharmaceutical, and Joincare Pharmaceutical each have two varieties listed.