Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-04-21
Posting Date:2023-04-21 Views:
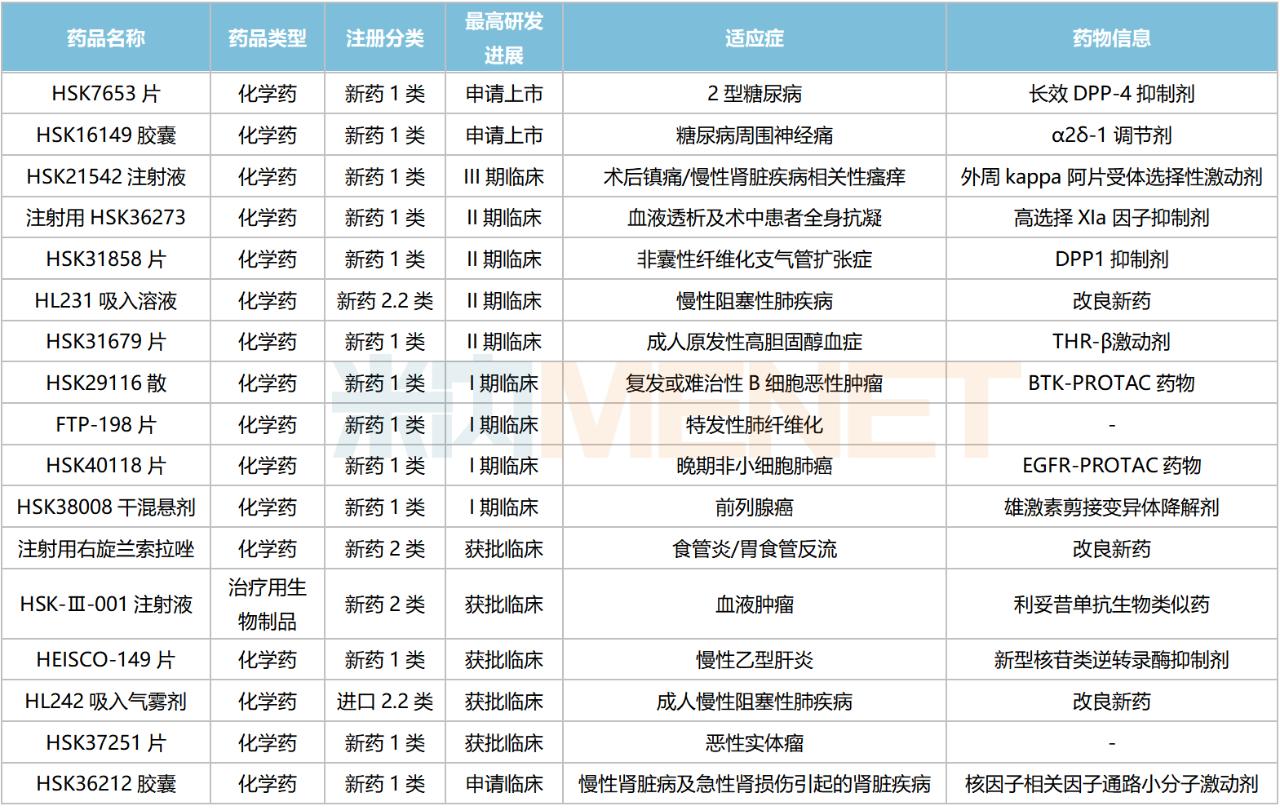
Views: Recently, Haisco has made new progress in its Class 1 new drug R&D, with HSK37251 Tablets and HSK38008 Dry Suspension receiving clinical trial approval for the first time. Currently, Haisco has 17 innovative drugs (13 Class 1 new drugs) at or beyond the stage of clinical trial application, focusing on therapeutic areas such as anesthesia and analgesia, oncology, respiratory, and digestive diseases. Its new anesthesia drugs are rapidly gaining market share, with two Class 1 analgesic drugs expected to be launched soon. The company’s innovative oncology platform continues to gain momentum, with its first biologic targeting a RMB 5.5 billion drug category. Several inhalation drugs are also in the pipeline, and its first new diabetes drug has already been submitted for marketing approval.
Sales of Anesthesia New Drugs Soar, Two Class 1 Analgesics Await Launch
Nervous system drugs are a key focus area for Haisco, concentrated in the two sub-categories of anesthesia and analgesia. According to Minenet data, the sales scale of chemical drugs for the nervous system in China’s public medical institutions (urban public hospitals, county-level public hospitals, community health centers, and township health centers) exceeded RMB 100 billion in 2021.
Haisco’s Nervous System Drugs (New Drugs) R&D Status in China
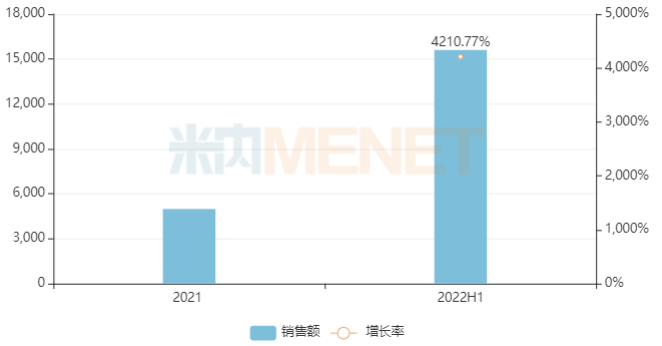
The company’s first Class 1 new drug, Ciprofol Injection, was first approved for marketing at the end of 2020, with its indications for sedation during gastrointestinal endoscopy and general anesthesia induction included in the national medical insurance catalog (Category B) through negotiation. According to Minenet data, sales of Ciprofol Injection in China’s public medical institutions in the first half of 2022 exceeded RMB 150 million, an increase of 4,210.77% compared to the same period last year.
Sales of Ciprofol Injection in China’s Public Medical Institutions (Unit: RMB 10,000)
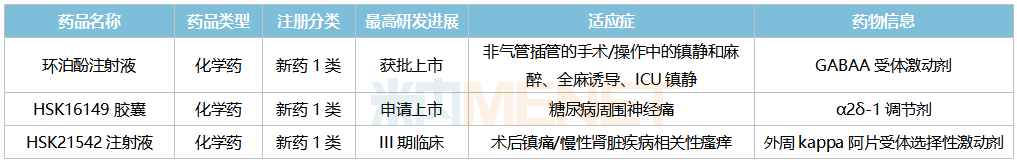
HSK16149 Capsules, an α2δ-1 modulator, are a me-better drug of pregabalin, featuring high target selectivity, potent and long-lasting analgesia, and minimal central side effects. An NDA has been submitted for its diabetic peripheral neuropathic pain indication, while Phase III clinical trials are ongoing for indications such as postherpetic neuralgia. Currently, no drug is approved for diabetic peripheral neuropathic pain in China, and HSK16149 is expected to fill this market gap.
HSK21542 Injection is a selective agonist of the peripheral kappa opioid receptor, offering potent and long-lasting analgesic effects. Its indications for postoperative analgesia and pruritus associated with chronic kidney disease have both entered Phase III clinical trials.
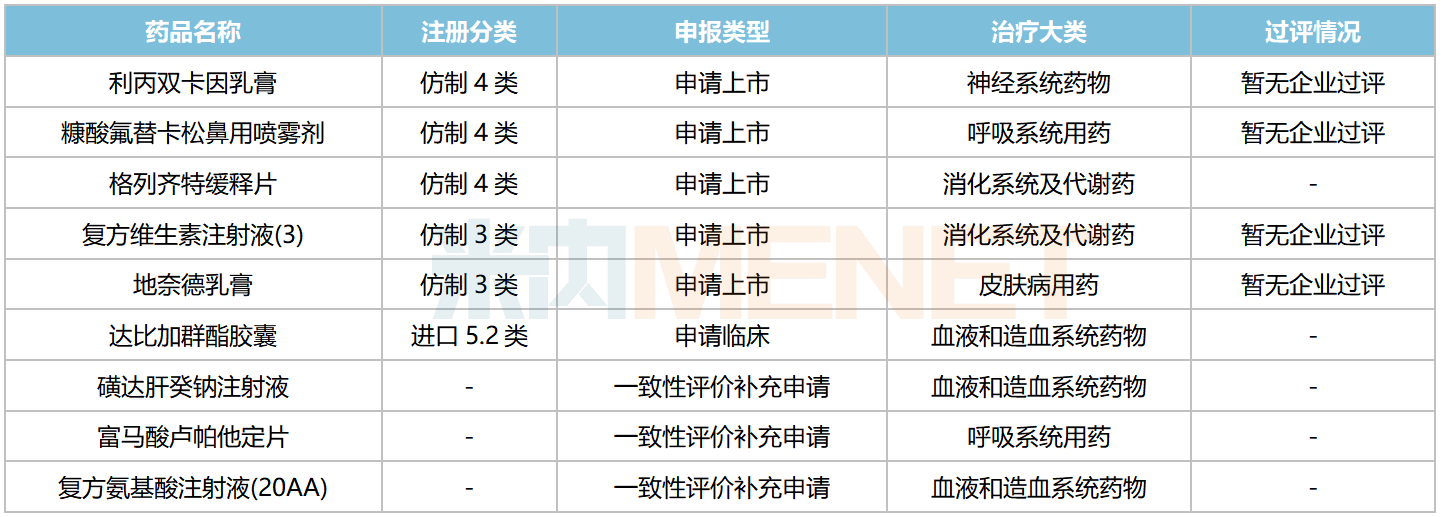
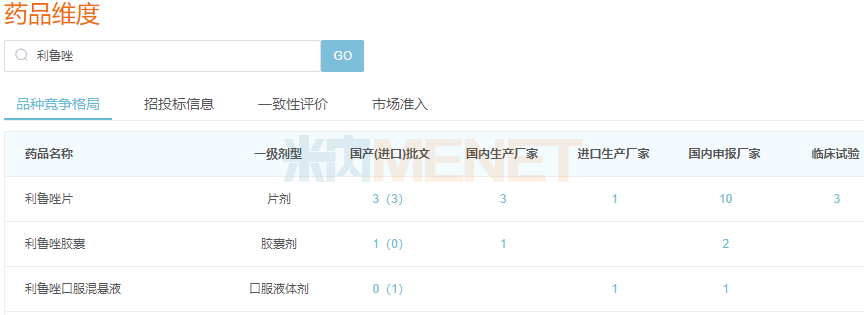
In addition to independent R&D, Haisco also introduces innovative drugs through licensing. In March 2022, Haisco licensed Exservan (riluzole oral film) from Aquestive. This is a glutamate excitotoxicity antagonist used to treat amyotrophic lateral sclerosis (ALS). Compared with traditional formulations, Exservan provides a meaningful treatment option for ALS patients with dysphagia or who cannot swallow.
Riluzole Formulations Approved for Marketing in China
Innovative Platform Continues to Gain Momentum, First Biologic Targets RMB 5.5 Billion Category
The Proteolysis Targeting Chimera (PROTAC) technology platform is one of the core platforms for Haisco’s innovative drug R&D. PROTAC is a technology developed based on the ubiquitin-proteasome system that uses small molecule compounds to induce the degradation of target proteins. This technology innovates at the drug design source, overcoming the development challenges of traditionally “undruggable” targets and redefining the capabilities of small molecule drugs. It is considered a revolutionary technology in the biomedical field.
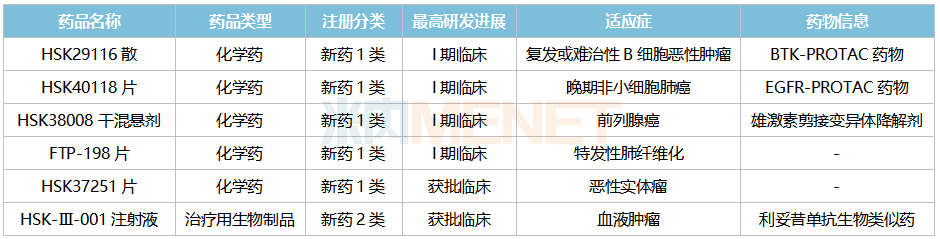
Haisco’s PROTAC platform currently has over 20 projects in its pipeline, primarily focused on oncology, including indications such as breast cancer, solid tumors, prostate cancer, and EGFR-mutant tumors. Among these, three Class 1 new drugs have entered Phase I clinical trials: HSK29116 Powder, HSK40118 Tablets, and HSK38008 Dry Suspension.
Haisco’s Antitumor and Immunomodulatory Agents (New Drugs) R&D Status in China
Source: Minenet Comprehensive Database
HSK29116 is the first PROTAC drug to enter clinical trials in China and is currently undergoing Phase I studies. This product has a dual antitumor mechanism (protein inhibition + protein degradation) and has the potential to become a first-in-class drug. Compared with marketed BTK inhibitors, HSK29116 is expected to achieve breakthroughs in target selectivity and BTK resistance mutations, offering new options for patients with B-cell malignancies.
HSK40118 Tablets are the world’s first PROTAC drug targeting EGFR, used to treat advanced non-small cell lung cancer. HSK38008 Dry Suspension is the world’s first oral androgen receptor splice variant degrader, designed to degrade androgen receptor splice variants through the proteasome pathway, blocking the transmission of the androgen receptor signaling pathway, thereby inhibiting the growth and proliferation of prostate cancer cells and achieving therapeutic goals for prostate cancer.
In the field of biologics, Haisco currently has only one antitumor drug in the clinical stage, HSK-III-001 Injection (a rituximab biosimilar). According to Minenet data, sales of rituximab across the three major terminals and six major markets in China exceeded RMB 5.5 billion in 2021.
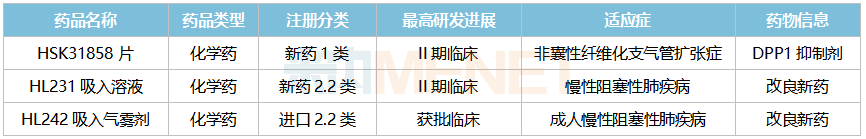
Several Inhalation Drugs Poised for Impact, Class 1 New Drug Aims for Domestic First
Haisco’s Respiratory System Drugs (New Drugs) R&D Status in China
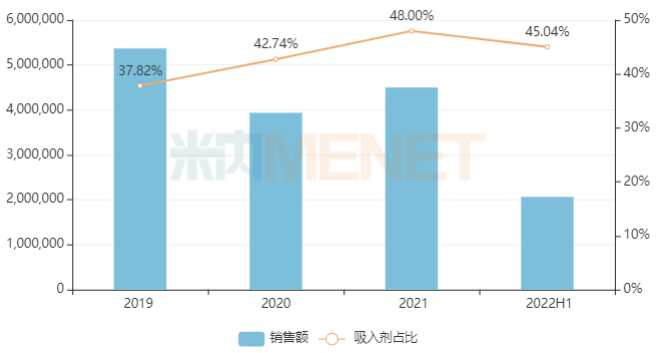
Data shows that the market size of chemical drugs for the respiratory system in China’s public medical institutions has fluctuated in recent years, with sales exceeding RMB 45 billion in 2021. In terms of administration route, inhalation drugs dominate, accounting for over 40% of the market share in recent years.
Sales of Chemical Drugs for Respiratory System in China’s Public Medical Institutions in Recent Years (Unit: RMB 10,000)
Leveraging its proprietary inhalation formulation technology platform, Haisco has developed multiple products including dry powder inhalers, metered-dose inhalers, and nebulizer solutions. Among these, the improved formulation new drug HL231 Inhalation Solution (Phase II clinical) and HL242 Inhalation Aerosol are both used to treat chronic obstructive pulmonary disease (COPD). In terms of generics, fluticasone furoate nasal spray has been submitted for marketing approval, and no generics for this product have been approved yet. Additionally, glycopyrronium dry powder inhaler and indacaterol maleate/glycopyrronium powder for inhalation have received clinical trial approvals and are currently undergoing clinical research.
Among Class 1 new drugs, HSK31858 Tablets, currently in Phase II clinical trials, are an oral, potent, and highly selective dipeptidyl peptidase 1 (DPP1) inhibitor. Currently, no drug targeting this mechanism has been approved globally. AstraZeneca’s INS1007 has entered Phase III clinical trials, while HSK31858 Tablets are the most advanced in the Chinese market.
First Diabetes Drug Targets Over RMB 4 Billion Market, Innovative High-End Formulation Licensed
Haisco’s Digestive System and Metabolic Drugs (New Drugs) R&D Status in China

Currently, Haisco has no approved diabetes-related products. Among its new drugs under development, HSK7653 Tablets have been submitted for NDA. This is a long-acting dipeptidyl peptidase-4 (DPP-4) inhibitor, with the potential to become the longest-acting oral diabetes drug globally. Currently, six DPP-4 inhibitors (excluding compound formulations) are approved for marketing in China, but no domestic Class 1 new drug has been approved.
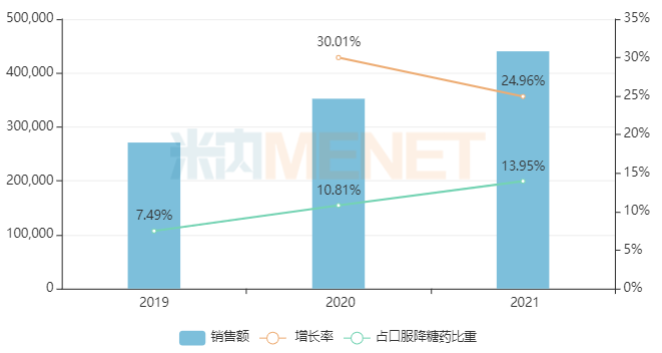
Leveraging advantages such as good safety and tolerability, no significant effect on body weight, and no increased risk of hypoglycemia, the market size of DPP-4 inhibitors across the three major terminals and six major markets in China has been growing at double-digit rates in recent years, with their share of oral diabetes drugs increasing year by year.
Sales of DPP-4 Inhibitors Across China’s Three Major Terminals and Six Major Markets in Recent Years (Unit: RMB 10,000)
In digestive system and metabolic drugs, Haisco has been深耕 in the antiemetic field for many years, with products such as Dolasetron Mesylate Injection and Palonosetron Hydrochloride Injection. In March 2021, the company invested $7.5 million to license Starton’s investigational drug, Olanzapine Transdermal Patch (STAR-OLZ), for the treatment of chemotherapy-induced nausea and vomiting. STAR-OLZ is a once-weekly transdermal formulation, and currently, no olanzapine transdermal patch has been approved globally.
Olanzapine Formulations Approved in China

Haisco’s New Drugs Under Development in China (Excluding Approved)
Haisco’s Consistency Evaluation Varieties Under Review