



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-04-28
Posting Date:2023-04-28 Views:
Views: Tasly’s 21 Exclusive Products Bring in RMB 8.5 Billion, Secures Two First-to-Market Generics
Tasly has been in the news for all the right reasons this year: in March, it became the third domestic company to obtain approval for Nicorandil for Injection; in April, it submitted a clinical application for Antiwei Granules, a Class 1 new TCM patent medicine, and was the first to file for generic approval of Baclofen Oral Solution. As a leader in modern TCM, Tasly has accelerated its innovation-driven transformation in recent years, continuously increasing its R&D investment ratio. Its new product pipeline spans modern TCM, biologics, and chemical drugs. Currently, the company has an R&D pipeline encompassing 92 products under development, including 41 Class 1 innovative drugs, essentially forming a virtuous cycle of “one batch launched, one batch in clinical trials, one batch in development.” The company is also actively participating in major industry initiatives such as national centralized procurement and medical insurance negotiations, achieving remarkable results.
Solid Leadership in Modern TCM, 21 Exclusive Products Generate RMB 8.5 Billion in Sales
Founded in 1994, Tasly initially had only one product, Compound Danshen Dripping Pills. Since then, the company has focused on precise innovation in major therapeutic areas such as cardiovascular and cerebrovascular diseases, digestive metabolism, and oncology and immunology, forming a rich portfolio of modern TCM, chemical drugs, and innovative biologics.
Data shows that Tasly currently has 21 exclusive products, including 16 TCM patent medicines, 10 of which are nationally protected TCM varieties, 4 chemical drugs, and 1 biologic. The total sales of these exclusive products across the three major terminals and six major markets in China in 2021 exceeded RMB 8.5 billion.
Table 1: Tasly’s Exclusive Products
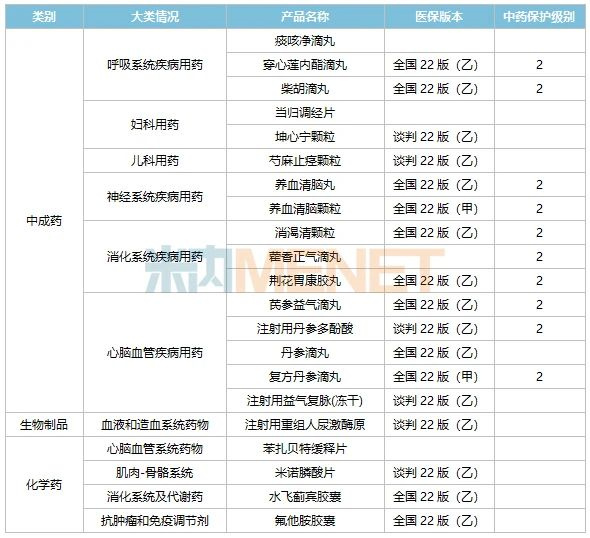
Note: National TCM protection statistics include varieties whose protection period has expired.
Tasly continues to deepen its presence in the TCM patent medicine market, with several exclusive products holding leading market positions. Nine exclusive TCM patent medicines—Compound Danshen Dripping Pills, Yangxue Qingnao Granules, Qishen Yiqi Dripping Pills, Yiqi Fumai for Injection (lyophilized), Salvianolic Acid for Injection, Yangxue Qingnao Pills, Andrographolide Dripping Pills, Huoxiang Zhengqi Dripping Pills, and Jinghua Weikang Capsules—each achieved sales exceeding RMB 100 million across the three major terminals and six major markets in China in 2021.
In the first half of 2022, within China’s public medical institutions (urban public hospitals, county-level public hospitals, community health centers, and township health centers), Tasly’s Compound Danshen Dripping Pills and Qishen Yiqi Dripping Pills ranked as the No. 1 and No. 11 oral cardiovascular drug brands, respectively. Yiqi Fumai for Injection (lyophilized) ranked as the No. 11 injectable cardiovascular drug brand. Salvianolic Acid for Injection ranked as the No. 9 injectable cerebrovascular drug brand. Yangxue Qingnao Granules and Yangxue Qingnao Pills ranked as the No. 1 and No. 2 oral headache (migraine) drug brands, respectively. Jinghua Weikang Capsules ranked as the No. 13 oral stomach remedy (gastritis, ulcers) brand.
In China’s urban physical pharmacy terminal, Tasly’s Andrographolide Dripping Pills is expected to rank as the No. 5 oral antipyretic and detoxifying drug brand in 2022, while Huoxiang Zhengqi Dripping Pills is expected to rank as the No. 2 oral anti- summer-heat drug brand.
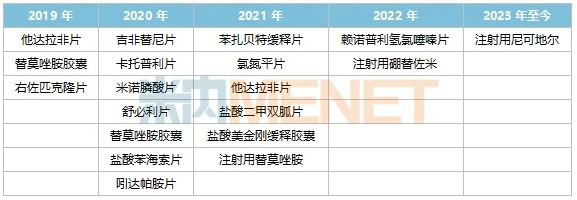
Figure 1: TCM Patent Medicines Approved by Tasly from 2019 to Date
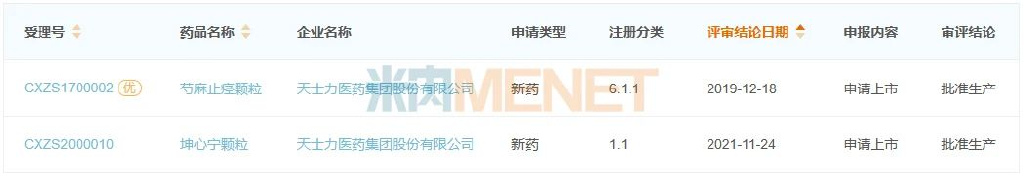
In 2019, the company obtained approval for Shaoma Zhijing Granules, a pediatric drug. In 2021, Kunxinning Granules, a gynecological drug, was approved. The approval of these two new TCM drugs has provided new momentum for the company’s performance growth.
In the chemical drug and biologics market, Tasly’s exclusive products, Silybin Capsules and Recombinant Human Prourokinase for Injection, achieved sales of RMB 790 million and RMB 290 million, respectively, across the three major terminals and six major markets in China in 2021. In the first half of 2022, Silybin Capsules ranked as the No. 4 oral drug brand for biliary and liver diseases in China’s public medical institutions.
Since 2019, the company has successively approved 8 new chemical drugs. Among these, Minodronic Acid Tablets (approved in 2020) and Bezafibrate Sustained-Release Tablets (approved in 2021) were first-to-market generics in China and remain exclusive products. As the company increases its R&D for new chemical drugs and biologics, its ability to develop generic drugs is also continuously improving. Currently, the company has 5 new chemical drugs under review for marketing, and Tasly is poised to compete for domestic first-to-market status for these products.
41 Class 1 New Drugs Shine Bright! 3 New TCM Drugs and 1 New Chemical Drug in the Pipeline
Figure 2: Tasly’s R&D Investment in the Past Three Years (Unit: RMB 100 million)
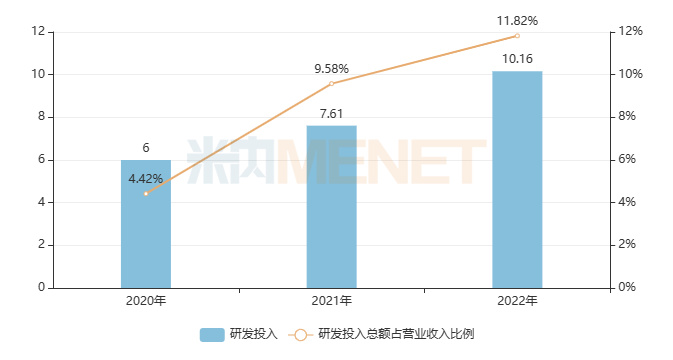
Earlier, Tasly released its 2022 annual report, showing R&D investment of RMB 1.016 billion, accounting for 11.82% of its operating revenue. The company adheres to an innovation-driven strategy, continuously increasing R&D investment. Through its “Four-in-One” R&D model, it has an R&D pipeline encompassing 92 products under development, including 41 Class 1 innovative drugs. Among these, 39 have entered clinical trials, and 24 are in Phase II or Phase III clinical studies.
Tasly’s R&D for innovative TCM drugs and the secondary development of key varieties is progressing rapidly. In addition to its primary focus areas of cardiovascular and cerebrovascular diseases, digestive system, and nervous system, the company has also been building pipelines in gynecology, otorhinolaryngology, dermatology, respiratory system, musculoskeletal system, and pediatrics in recent years, which is expected to enter a period of fruitful outcomes.
Table 2: Selected Key New TCM Drugs from Tasly in Phase I Clinical Trials or Beyond
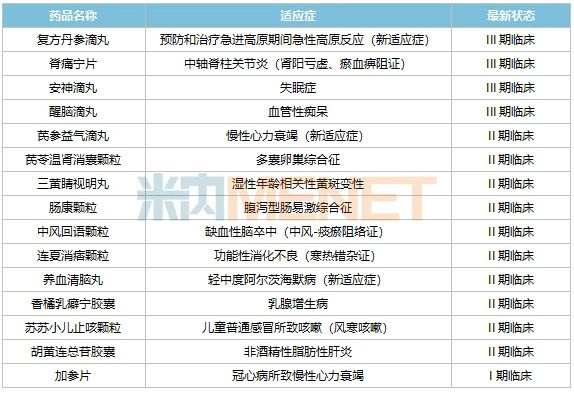
Based on current progress, Jitongning Tablets, Anshen Dripping Pills, and Xingnao Dripping Pills are in Phase III clinical trials, just one step away from applying for marketing approval. It is reported that Jitongning Tablets, derived from the hospital preparation Jitongning Capsules, is a pure TCM preparation developed based on years of clinical experience for the treatment of axial spondyloarthritis. Anshen Dripping Pills, also originating from a clinical empirical formula, is a pure TCM dripping pill preparation used to treat neurasthenia, insomnia with excessive dreaming, restlessness, and other symptoms.
In 2022, the company’s Class 1.1 new TCM drug, Jiuwei Huaban Pills, received clinical trial approval for indications of blood heat syndrome and blood stasis syndrome in moderate to severe plaque psoriasis. Qishen Yiqi Dripping Pills also received a clinical trial approval for a new indication, diabetic nephropathy. In 2023, the company filed for another Class 1.1 new TCM drug. According to data from the WHO International Clinical Trials Registry Platform, Antiwei Granules may be intended for the treatment of influenza, and its clinical application is currently under review.
Table 3: Selected Key New Chemical Drugs and Biologics from Tasly in Phase I Clinical Trials or Beyond
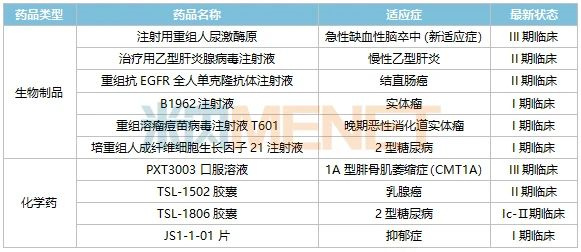
Tasly’s biologics R&D is progressing steadily in a tiered portfolio. Its chemical drug R&D adheres to the principle of “equal emphasis on innovation and generic development,” covering not only popular therapeutic areas such as oncology and diabetes but also including new drugs for rare diseases.
PXT3003 is a new chemical drug and the world’s first treatment for Charcot-Marie-Tooth disease type 1A (CMT1A). In 2017, Tasly invested in France’s Pharnext through equity and convertible bonds and established a joint venture in China, Tasly International Genetic Network Drug Innovation Center Co., Ltd. (with Tasly holding 65%), to license this product. According to the annual report, the new drug has completed enrollment of all patients in Phase III clinical trials.
Figure 3: Global R&D Progress of PXT3003
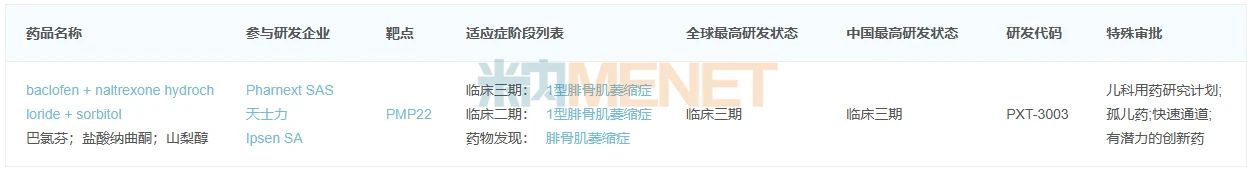
Source: Minenet Global New Drug R&D Data
New TCM Drugs and Exclusive Chemical Drugs Enter National Reimbursement; Market Share of Centralized Procurement Winners Soars
Earlier this year, the 2022 National Reimbursement Drug List (NRDL) negotiations were successfully completed. Tasly’s Class 1.1 new TCM drug Kunxinning Granules and its exclusive chemical drug Minodronic Acid Tablets were newly added to the NRDL, while Shaoma Zhijing Granules successfully renewed its contract. Products selected in the seventh round of national centralized procurement, such as Temozolomide for Injection and Memantine Hydrochloride Sustained-Release Capsules, were directly included. According to the company’s annual report, Tasly currently has over 160 products included in the 2022 version of the NRDL.
Table 4: Tasly’s Products That Have Passed Consistency Evaluation (Including Deemed Equivalents)
In addition to NRDL negotiations, consistency evaluation and national centralized procurement are also major industry events. To date, Tasly has 17 products that have passed or are deemed to have passed consistency evaluation. Among these, Bezafibrate Sustained-Release Tablets, Lisinopril Hydrochlorothiazide Tablets, Minodronic Acid Tablets, Sulpiride Tablets, Temozolomide Capsules, Trihexyphenidyl Hydrochloride Tablets, and Eszopiclone Tablets were the first to pass the evaluation.
Table 5: Changes in Market Share of Tasly’s Products Selected in National Centralized Procurement
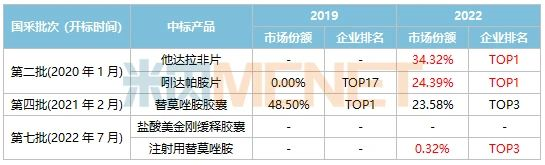
Source: Minenet Key Provincial & Municipal Public Hospital Drug Terminal Competitive Landscape
Tasly successfully won bids for a total of 5 products in the second, fourth, and seventh rounds of national centralized procurement. Among these, Tadalafil Tablets, Indapamide Tablets, and Temozolomide for Injection saw their market shares surge after the implementation of centralized procurement. In 2022, at the level of key provincial and municipal public hospitals, Tasly became the leading enterprise for Tadalafil Tablets and Indapamide Tablets. With the support of centralized procurement, the company’s chemical drugs are poised for new breakthroughs.
Conclusion
On April 25, Tasly released its first-quarter 2023 report, showing operating revenue of RMB 2.080 billion, a year-on-year increase of 10.92%, and net profit of RMB 266 million, a year-on-year increase of 148%. Multiple securities firm research reports indicate that Tasly’s performance is expected to significantly improve in 2023, marking a turning point and entering a new cycle of profitable growth.