



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-04-14
Posting Date:2023-04-14 Views:
Views: The relationship between medical insurance reimbursement standards and drug prices is an issue that enterprises must pay close attention to. The adjustment methods for the prices and reimbursement standards of non-selected products in centralized procurement, in particular, not only determine the reimbursement level of the relevant products but also indirectly affect their clinical competitiveness.
Hubei Province has been making frequent moves recently. Before the dust could settle on the draft of the national TCM patent medicine centralized procurement led by Hubei, the province subsequently initiated adjustments to the prices and medical insurance reimbursement standards for non-selected centralized procurement products.
The varieties being adjusted include:
Non-selected varieties from the first to fifth rounds of national centralized procurement, including a batch of original drugs and reference-listed drugs;
Varieties from Hubei provincial centralized procurement;
Non-selected varieties from the Hubei-led 19-province TCM patent medicine centralized procurement;
Non-selected varieties from the Chongqing-Hubei-Hainan-Qinghai-Ningxia-Xinjiang-Xinjiang Production and Construction Corps common drug alliance volume-based procurement.
On April 11, the Hubei Provincial Medical Insurance Bureau issued the “Notice on Adjusting the Price Information of Certain Listed Drugs” on the Hubei Provincial Medical Insurance Price and Bidding Procurement Management Service Network. The notice indicated that Hubei Province had reviewed and publicized certain listed drugs whose listed prices were voluntarily reduced by the manufacturers, and the new prices would be implemented in all public medical institutions across the province starting April 14.
A total of 21 listed drugs, involving 18 companies and 18 generic names, had their listed prices adjusted.
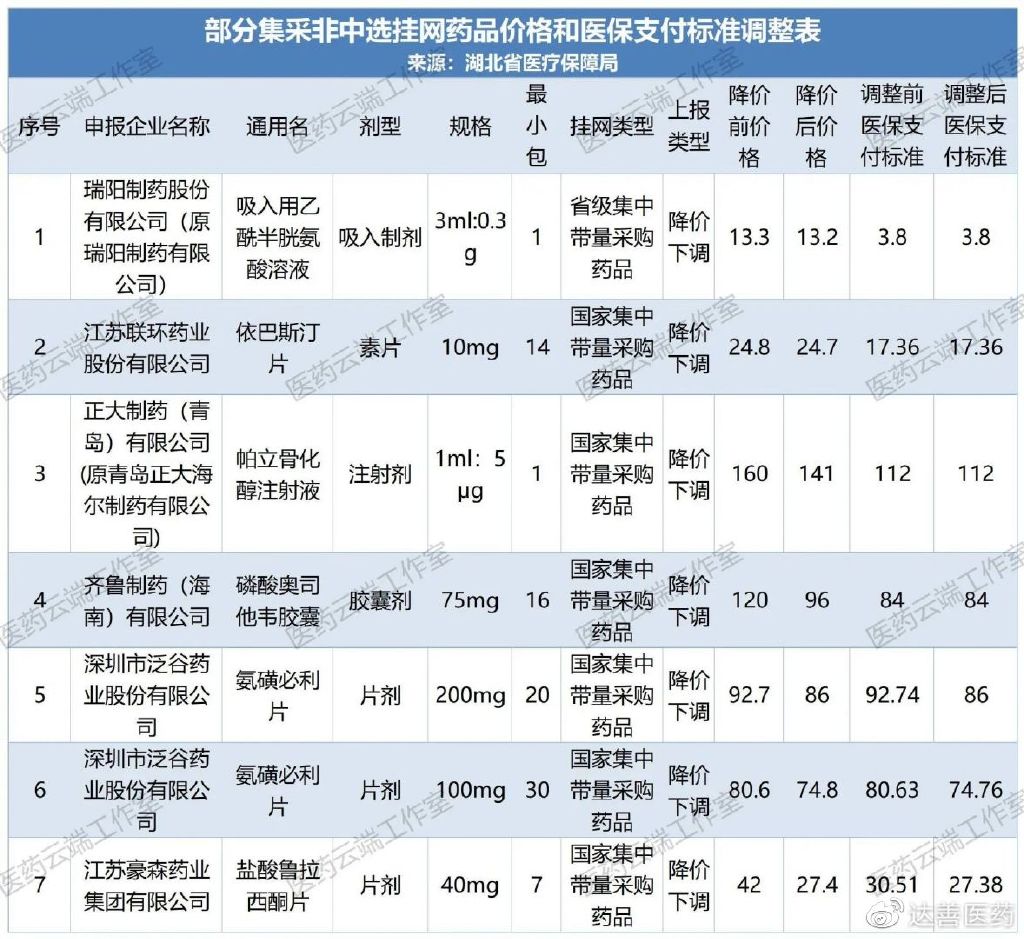
Among these, the adjustments to prices and medical insurance reimbursement standards for certain non-selected centralized procurement listed drugs are shown in the table below:
This round of adjustments to reimbursement standards involved 6 generic names and 6 manufacturers. The magnitude of reductions varied across different products, from the voluntary reduction of listed prices to the adjustment of medical insurance reimbursement standards.
For example, the price of Ruiyang’s Acetylcysteine Solution for Inhalation was slightly adjusted from RMB 13.3 to RMB 13.2, while the reimbursement standard remained unchanged at RMB 3.8, with the reimbursement portion accounting for only 28.79%. The price of Zhengda Pharmaceutical (Qingdao)’s Paricalcitol Injection was reduced from RMB 160 to RMB 141, a decrease of 11.88%, while the reimbursement standard remained unchanged at RMB 112, with the reimbursement portion accounting for over 79%.
According to the relevant regulations of national and provincial centralized procurement, the winning price in centralized procurement is coordinated with the reimbursement standard, meaning the reimbursement standard is the winning price. For non-selected products (those that were not selected or did not participate in centralized procurement), the reimbursement standard will also change along with price reductions based on a tiered approach or linkage to the national lowest price. Generally speaking, the reimbursement standard for non-selected products does not exceed the winning price.
Furthermore, on April 7, the Hubei Provincial Medical Insurance Bureau issued the “Notice on Publicizing Information such as Proposed Listed Prices for Non-Selected Varieties from the First to Fifth Rounds of National Centralized Drug Procurement.” The public notice period was from April 8 to April 12, 2023. The requirement was to carry out related work for non-selected varieties under the same generic names from the first to fifth rounds of national centralized drug procurement, including drug codes, consistency evaluation attributes, adjustment of listed prices, and suspension of listing.
According to the provisions and requirements of the relevant documents, for non-selected drugs that are original drugs, reference-listed drugs, or generic drugs that have passed consistency evaluation, the upper limit is the lowest current winning/listing price on provincial platforms nationwide. Enterprises independently declare their listing prices and are encouraged to proactively reduce prices. For drugs without a provincial platform winning/listing price, the lowest price among other manufacturers for the same specification is used as their listing price.

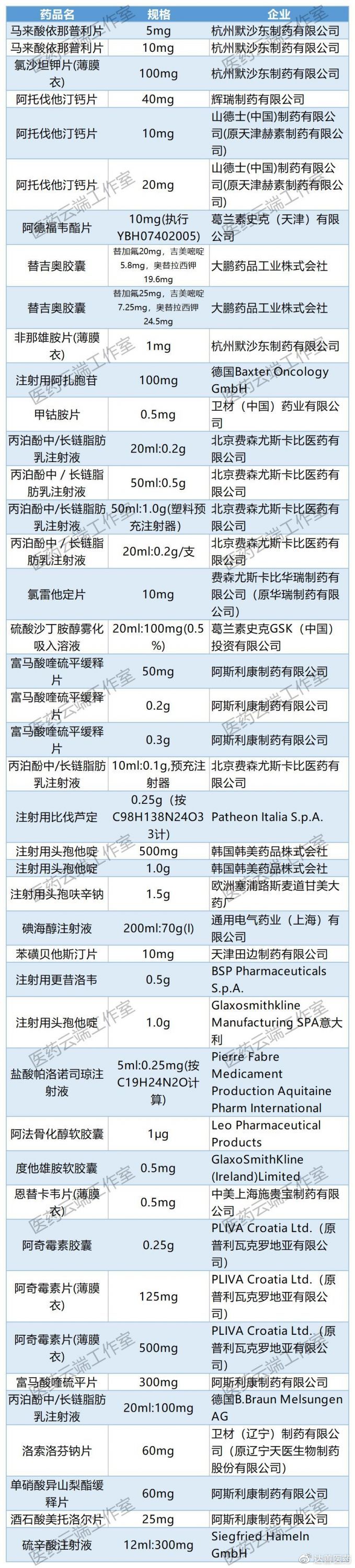
In Appendix 6, “Proposed Suspension List for Non-Selected Varieties from the First to Fifth Rounds of National Centralized Drug Procurement,” 880 specifications from 388 companies are proposed for suspension. This includes many products from foreign enterprises, including original drugs and reference-listed drugs, such as Pfizer’s Atorvastatin Calcium Tablets, which were not selected in the 4+7 centralized procurement. As shown in the table below:
On April 10, the Hubei Provincial Medical Insurance Bureau issued the “Notice on Carrying Out Information Declaration and Price Adjustments for Non-Selected Varieties in Interprovincial Alliance Centralized Volume-Based Procurement Projects such as Chongqing Common Drugs and the First Batch of TCM Patent Medicines.” Non-selected varieties from these alliance procurement projects for chemical drugs and TCM patent medicines will also face price adjustments. Manufacturers were required to complete information maintenance and listing price adjustments by 24:00 on April 13. Drugs failing to apply for price adjustments within the specified timeframe will have their listings suspended.
According to the relevant document requirements for the listing prices and medical insurance reimbursement standards of non-selected varieties in Hubei’s TCM patent medicine centralized procurement, such varieties need to link to the national lowest listing/procurement price, and the medical insurance reimbursement standard is to be reduced based on the average price reduction of selected varieties.
In September 2021, Hubei Province issued the “Notice on the Work of Admission and Dynamic Adjustment for Drug Procurement in Public Medical Institutions” (E Yi Mi Fa [2021] No. 46). Hubei Province included all drugs (excluding those otherwise stipulated by the state and province) within the scope of admission listing and dynamic adjustment management. All drugs are classified according to 9 standards, and corresponding listing and adjustment rules are formulated based on their classification.
The pricing rules for the 9 categories of drugs, aside from implementing the listing price policies explicitly stipulated by the state and province, generally follow the principle of setting prices based on the lowest listed price limit on provincial-level procurement platforms nationwide. A price linkage mechanism is also established to ensure that the listed prices of all drugs remain at a consistently low level nationwide.