



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-04-07
Posting Date:2023-04-07 Views:
Views: Within two weeks, following Guangdong, Inner Mongolia, and Hubei’s issuance of their provincial lists of key monitored rational drug use drugs (second batch), ten more provinces, including Heilongjiang, Jiangsu, and Shaanxi, have successively rolled out their monitoring frameworks. Some provinces have added drugs to the national second‑batch list, expanding the scope and imposing stricter requirements. (Click to read: “Second Batch of National Key Monitoring Lists Rolled Out”)
On March 31, the Tianjin Municipal Health Commission issued the “Notice on Further Strengthening the Clinical Use Management of Key Monitored Drugs in Medical Institutions,” releasing the “Second Batch of Tianjin Key Monitored Rational Drug Use Drugs,” which includes 43 drugs—13 more than the national version.
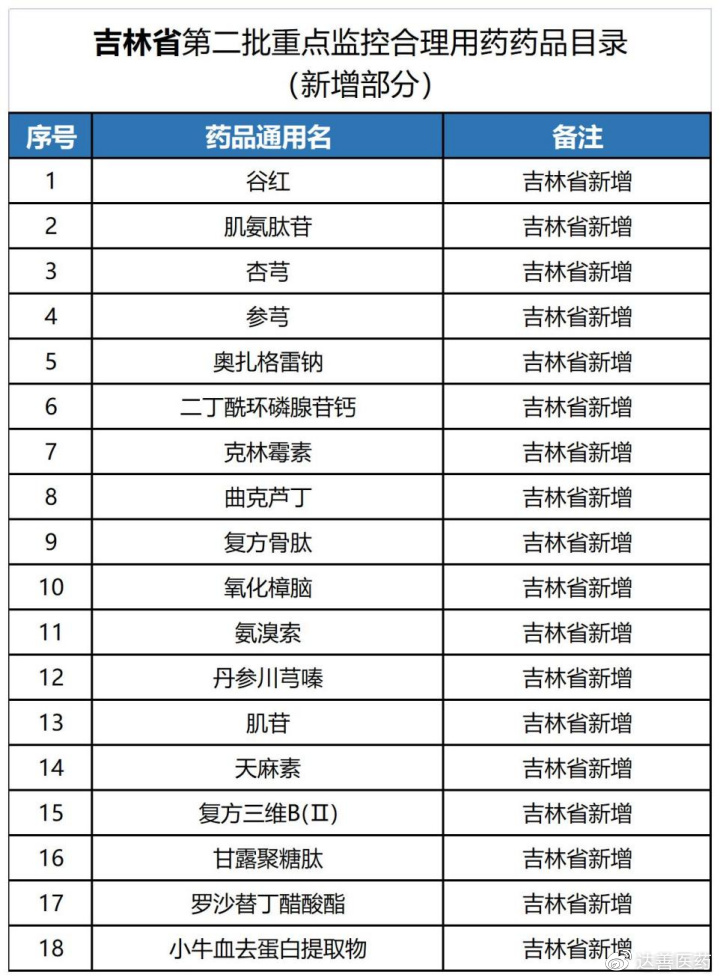
On March 27, the Jilin Provincial Health Commission announced its list, adding 18 drugs to the national second‑batch version—the largest provincial addition so far.
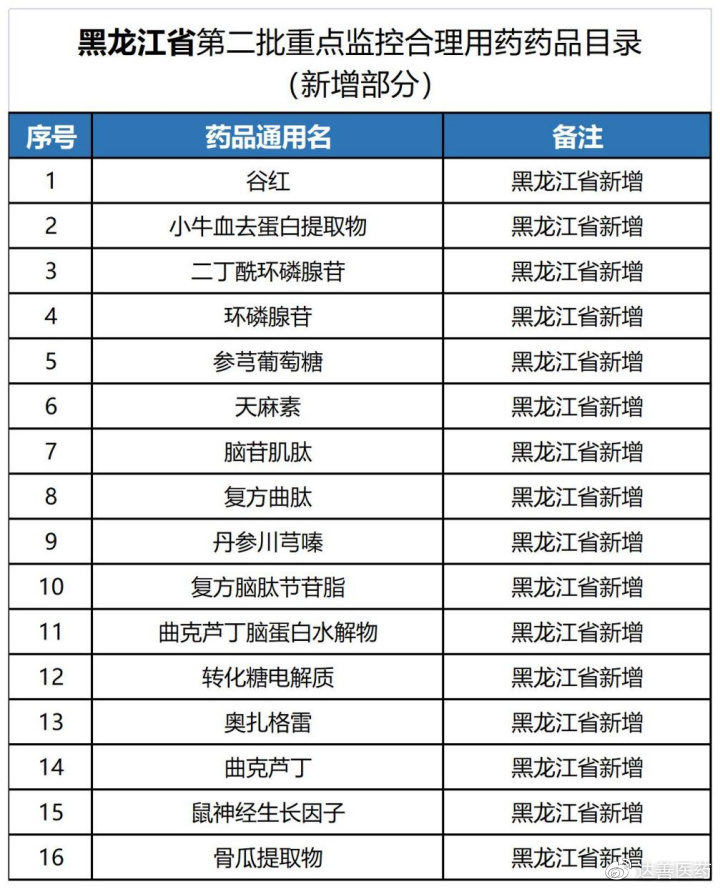
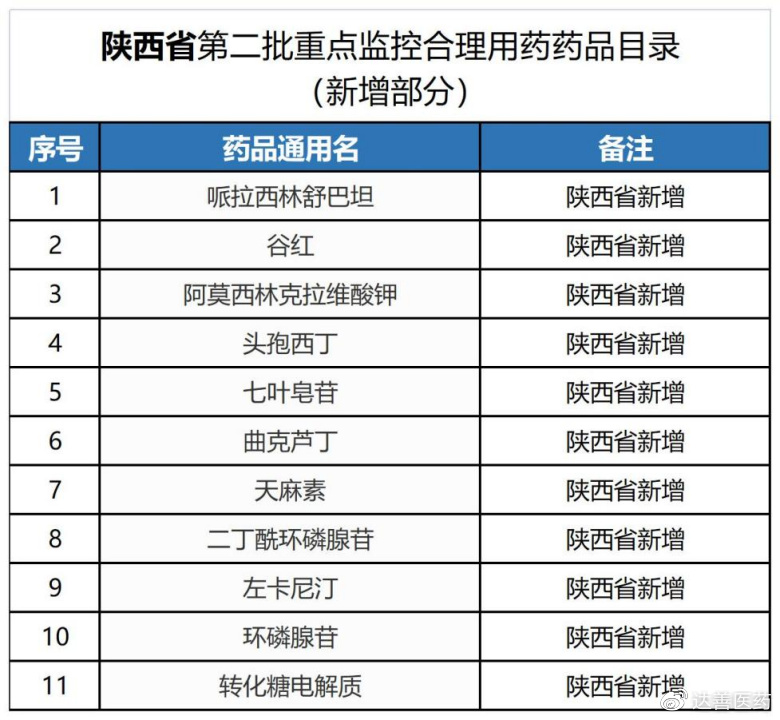
Additionally, Heilongjiang added 16 drugs, explicitly stating that patient needs should not be compromised by simplistic “drug suspension” management. Shaanxi added 11 drugs, six of which are from the eighth round of centralized procurement. As more provinces follow suit and expand their monitoring lists, how to supervise and manage these key drugs has become a focal issue.
Provinces Add “Centralized Procurement Heavy Hitters”
Stricter Requirements
On January 13, the National Health Commission released the “Second Batch of National Key Monitored Rational Drug Use Drugs,” which includes 30 major drugs such as omeprazole, human albumin, cefoperazone sodium/sulbactam sodium, edaravone, and ginkgo biloba extract. Seven of these were from the first batch. For first‑batch drugs not included in the second batch, local health administrative departments must continue monitoring for at least one year; after that, monitoring may be discontinued based on rational use assessments.
Including the 20 drugs from the first batch, a total of 43 drugs will now be under national key monitoring.
Jilin’s list adds 18 drugs, including gulhong, ozagrel sodium, troxerutin, ambroxol, etc. The provincial health commission requires medical institutions to finalize their internal lists by April 20.
Notably, gulhong and deproteinized calf serum, not previously included in Hubei’s monitoring list, were added by Jilin. This shows that provinces are tailoring their lists based on factors such as irrational use, expenditure, and clinical value, making local requirements stricter than the national baseline.
Heilongjiang added 16 drugs, requiring medical institutions to develop their own lists based on the provincial list and submit them for record and public disclosure by April 21.
Notably, Heilongjiang emphasized scientific, refined management to promote rational use, cautioning against the simplistic approach of “suspending drugs” that could affect patient needs.
Shaanxi added 11 drugs to the national list of 30. Among these, six are from the eighth round of centralized procurement: amoxicillin clavulanate, cefoxitin, levocarnitine, cefoperazone sulbactam, piperacillin tazobactam, and cefotaxime. This inclusion suggests that even drugs selected in centralized procurement can be subject to irrational use or high expenditure.
Industry observers suggest that these drugs have stable, substantial clinical demand and have already undergone price reductions. Placing them under key monitoring may be primarily aimed at ensuring rational use and curbing misuse. While centralized procurement has lowered drug prices, it has not solved the issue of irrational use, and may even have inadvertently contributed to it. This emerging trend, reported by local medical institutions, warrants attention from regulatory authorities.
Pressure Rises for Key Monitored Drugs
How Will Hospitals Manage?
Beyond adding drugs to the national list, provinces are also implementing self‑imposed stricter measures:
Shandong’s second‑batch list includes 30 drugs, while Zibo City’s third‑batch list includes 36.
Guangxi forwarded the national list without additions but required cities to develop their own lists within 30 days, with hospital lists containing no fewer drugs than the city‑level list. This indicates that at each level, medical institutions will further refine their own key monitoring lists, resulting in more monitored drugs at lower administrative levels.
With intensified monitoring, the market performance of these drugs is drawing increasing attention.
Industry insiders note that to expand usage within institutions under stricter compliance and rational use requirements, products must rely on solid evidence‑based medicine, inclusion in clinical pathways, guidelines, or expert consensus. Regulatory authorities require hospitals to strengthen prescription reviews, conduct prescription comments and disclosures, and implement measures for unusual prescriptions—essentially aligning with the clinical‑value‑oriented market reshuffling.
Notably, many drugs on the second‑batch list are first‑line treatments, reflecting stronger efforts to curb drug misuse. Hospitals are expected to monitor all dosage forms of these drugs rather than focusing on specific forms.
Provincial health commissions have outlined key management requirements: develop clinical guidelines for key monitored drugs, clearly define clinical use principles and conditions, strictly control indications, and avoid arbitrarily expanding indications or altering treatment duration, administration routes, or dosage. For drugs with prominent misuse issues, measures such as ranking notifications and corrective actions will be applied to gradually improve rational use.
Tianjin’s health commission explicitly requires that the clinical use of drugs on the list be incorporated into rational use management and public hospital performance evaluations. Prescription reviews and comments will be continuously strengthened, with monthly monitoring, analysis, and assessment of drug use to identify and address irrational use promptly.
Jilin’s health commission mandates that the use of key monitored drugs be included as an important evaluation indicator in hospital accreditation, public hospital performance assessments, and rational use evaluations. Health administrative departments must strengthen supervision of these drugs and integrate their clinical use into institutional assessments.
Notably, Inner Mongolia’s health commission linked key monitored drug use directly to anti‑corruption efforts, specifying that key drug use should be monitored as part of institutional, medical personnel, and medical conduct oversight to prevent integrity risks in procurement and use.
Market observers note that while national lists and policies set principles and directions for rational use, implementation and refinement will require proactive efforts from local authorities and medical institutions to develop effective practices.