



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-02-03
Posting Date:2023-02-03 Views:
Views: Optimism amid the frenzy of pharmaceutical innovation and pessimism during the industry downturn both impede objective strategic decision-making. Therefore, it is essential to view the current industry winter and the upcoming new wave of innovation from industrial, historical, and global perspectives, cut through the industry fog, gain insights into the industrial future, and plan development paths with the end in mind.
The Previous Innovation Frenzy Was Undoubtedly Inflated
The last round of biomedical innovation was fueled by a large number of scientists returning to China to innovate and launch startups. Starting around 2010, driven by China’s shift to innovation-driven development, the continuous optimization of the business environment, and evolving international dynamics, over 1,400 Chinese life scientists returned to start businesses, and biotech companies mushroomed across the country. According to a BCG survey, China currently has approximately 1,600 biotech firms.
Joining the second echelon of global innovation cannot conceal its relatively weak foundation. In 2019, a McKinsey report stated that China had risen from the third to the second echelon in global pharmaceutical innovation. McKinsey’s definition of global innovation tiers is mainly determined by “the number of pre-market clinical R&D trials” and “the number of newly launched drugs”. However, a considerable portion of China’s innovative drugs are fast-follow me-too drugs, with relatively few first-in-class (FIC) and best-in-class (BIC) drugs, and extremely few potential international blockbuster drugs that could rewrite diagnosis and treatment guidelines. Meanwhile, most innovative pharmaceutical companies contributing to new drugs are relatively newly established, with weak accumulation of technology, experience, and capital, and basic life science research provides relatively weak support for pharmaceutical innovation.
Hidden bubbles emerged amid the industry’s prosperity. As China’s biomedical industry required extensive learning and exploration, many enterprises had imperfect core technologies, naturally resulting in a large number of fast-follow me-too products, few FIC and BIC drugs, and extremely few potential international blockbuster drugs that could rewrite diagnosis and treatment guidelines. There was massive redundant development of popular low-barrier targets, risk capital scrambled for projects, secondary market valuations far exceeded those of the world’s top 20 enterprises, and the industry was brewing numerous bubbles amid its boom.
The Industry Winter Is a Self-Repair of Pharmaceutical Innovation
The normal “New Clinical Value Policy” triggered the industry’s downturn. Strictly speaking, the November 2021 New Clinical Value Policy was not new; it originated from the Drug Administration Law and is consistent with ICH requirements. However, combined with the China-US strategic competition and other events, the pharmaceutical industry shifted from prosperity to downturn. Many innovative drugs failed to meet commercial expectations, secondary market stock prices plummeted, primary market investment became cautious, and a considerable number of biotechs faced cash flow shortages and unsustainable dilemmas.
The cycle of seasons is also a natural law of biomedical industry development. Excessive redundant target development, unreasonable high expectations, and inflated valuations are ultimately unsustainable. Therefore, the industry’s shift from prosperity to recession and downturn is a natural adjustment, and a necessary one to welcome the next spring.
The pharmaceutical winter has positive implications. It is naturally cruel for enterprises unprepared for the downturn, but for those prepared or capable of recovery, most are reflecting on the irrationalities of the previous boom, identifying key success factors for enterprises and products under high-quality development, and building internal strengths for the next wave of innovation. Meanwhile, the survival of the fittest among some enterprises and products frees up valuable capital and market space for high-value new drugs, and industrial policies will tend to support innovative drugs with more significant value. Thus, in the winter, the survivors prevail, and what does not kill you makes you stronger.
If winter comes, can spring be far behind?
The next wave of innovation is gathering momentum. The wave of pharmaceutical innovation driven by explosive breakthroughs in genomics and proteomics is still on the way. It is reported that humans currently know more than 20,000 targets, of which only over 600 have drugs in clinical trials or on the market, and target development is still in the initial stage. On the other hand, with technological breakthroughs, innovative technologies such as CAR-T/NK, oncolytic viruses, stem cells, and nucleic acid drugs are emerging and maturing. More and more gene therapy and cell therapy products have entered clinical development and been approved for marketing, and synthetic biology has also made major breakthroughs. New mechanisms, new targets, and new technologies are on the eve of a new round of explosion.
Innovative pharmaceutical companies tempered by the winter have become more steady and robust. Those that have survived the industry cycles have not only gained clinical, capital, and social recognition for the clinical and commercial value of their product pipelines but also matured in technology. More importantly, most executive teams of innovative pharmaceutical companies are more committed to pursuing significant clinical value of products, focusing more on technology accumulation and breakthroughs, international development, and have a greater awe for the risks of innovative drug development.
China’s pharmaceutical innovation environment will keep improving. Amid the accelerated evolution of the once-in-a-century global changes, China’s transformation from a major pharmaceutical country to a pharmaceutical power resonates with the great rejuvenation of the Chinese nation. Against the backdrop of national expectations for the pharmaceutical industry to grow stronger, medical insurance continuously eliminating drugs without significant clinical value advantages to make room for pharmaceutical innovation, and the deepening of other innovation-friendly policies, China’s pharmaceutical innovation environment will become better, attracting more Chinese scientists to return for development.
Rationally Understand the Historical Stage of China’s Pharmaceutical Innovation
China’s pharmaceutical industry is still in its adolescence. In a history spanning centuries, China’s local pharmaceutical innovation has only developed for more than 30 years from germination to growth. In terms of enterprise accumulation, talent cultivation, technology accumulation, policy environment, and basic research, it is still immature compared with established biomedical powers such as Europe and the United States, and lags behind Japan, which started earlier, in industrial depth. Historically, China’s biomedical innovation is still in its adolescence.
Thorns cannot obscure the bright prospects. Opportunities and accumulation determine the industrial development prospects. Although pharmaceutical innovation is highly risky, some countries are unwilling to see China’s biomedical industry upgrade and compete for their market share, and the current innovation environment still has many shortcomings, the new wave of innovation is gathering momentum. China has built a solid foundation for pharmaceutical innovation, and with the deepening of global changes, the rise of China’s biomedical industry is an irresistible trend.
The era calls for high-quality development of the pharmaceutical industry. In 2017, the 19th National Congress of the Communist Party of China first proposed the new concept of high-quality development, which is not only a clarion call for China to move from “becoming prosperous” to “becoming strong” but also a summons for China to transform from a major pharmaceutical country to a pharmaceutical power. The 13th Five-Year Plan basically ended the extensive development stage of China’s pharmaceutical industry and accelerated its progress toward high-quality development. The overall tone of high-quality development requires Chinese enterprises to develop blockbuster innovative drugs with significant clinical value that can stand international tests, and for China to foster a group of pharmaceutical enterprises among the world’s top 30.
The Pharmaceutical Sector Is Striving For Progress and Forging Ahead
A batch of innovative drugs expected to rewrite relevant diagnosis and treatment guidelines are being accelerated. From the pipeline development of enterprises such as BeiGene, Hutchison Medipharma, Ascentage Pharma, Legend Biotech, CStone Pharmaceuticals, and Rongchang Biologics, more and more enterprises take addressing unmet clinical needs and developing new drugs with significant clinical value as the primary orientation of product development. They explore the optimal molecular structures and positioning in their respective fields and verify clinical value, often rewriting relevant diagnosis and treatment guidelines upon product launch, creating significant value for clinics and patients.
The internationalization of Chinese pharmaceutical companies is unfolding. According to incomplete statistics, Chinese pharmaceutical companies concluded at least 44 cross-border License-out transactions in 2022, with the highest single transaction value reaching nearly 10 billion US dollars. Innovative drugs from BeiGene, Hengrui, Jacobio, Akeso, Ascentage, Junshi, Rongchang and other enterprises are accelerating the layout of overseas clinical trials and exploring international development models.
Number of Overseas Clinical Trials of Chinese Enterprises in the Past Decade
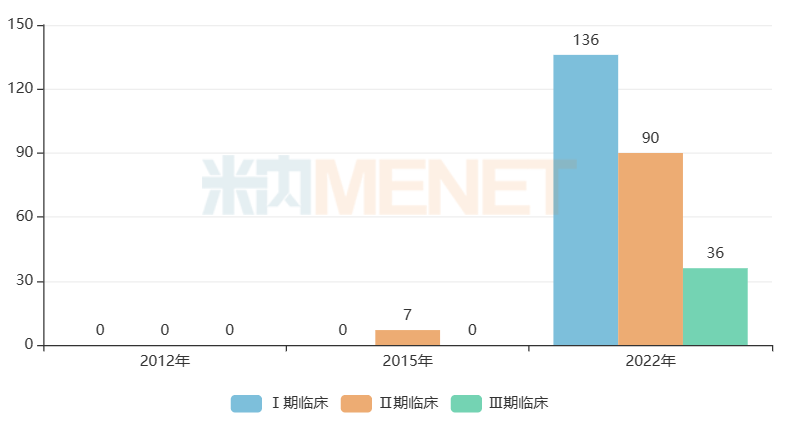
Source: Menet Global Drug R&D Database
A group of pharmaceutical companies are expected to gradually rank among the world’s top 30. As leading pharmaceutical enterprises led by Hengrui, Kelun, Sino Biopharm, Fosun accelerate innovation transformation and internationalization, and continuously enrich product pipelines expected to foster blockbusters; as innovative pharmaceutical companies such as BeiGene, Junshi, Hutchison, and CStone continuously layout potential blockbuster new drugs expected to rewrite diagnosis and treatment guidelines; and as the new wave of innovation approaches, a group of Chinese innovative pharmaceutical companies will gradually rank among the world’s top 30 in the future.
The institutional mechanism of basic life science research is continuously improving. At the national level, the General Secretary emphasized that Chinese science and technology workers should “aim at the frontiers of global science and technology, serve the main economic battlefield, meet the country’s major needs, and safeguard people’s life and health”. The national major R&D programs fully implement the “Ranking and Leading” mechanism to optimize the scientific and technological innovation management system. At the social level, a group of far-sighted enterprises have initiated and supported basic research programs such as the “New Cornerstone Investigator Program”, known as the Chinese version of the “HHMI Program”. Focusing on cutting-edge science and encouraging free exploration, it is expected to provide an outstanding source of innovation for pharmaceutical innovation at the source.
The innovation ecosystem is continuously optimized. The tone of high-quality development is promoting various CXO institutions to leverage their strengths, enabling clinical trial institutions to resonate with the industry, making pharmaceutical regulation more scientific, increasing innovation support, facilitating basic research to achieve major discoveries, and supporting basic and industrial research through “Double First-Class” talent training. Together with pharmaceutical enterprises, a strong and mutually supportive innovation ecosystem has been formed. Opportunities and challenges coexist. The adolescent Chinese pharmaceutical industry has boundless prospects and will surely experience trials and hardships. With the growth of China’s biomedical industry, more major achievements are worth looking forward to in the future!