



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-01-06
Posting Date:2023-01-06 Views:
Views: On January 5, the 2022 National Medical Insurance On-site Negotiations, postponed for over a month, officially commenced today. In contrast to the cold winter in Beijing, the atmosphere at the negotiation site was notably enthusiastic: the entrance to the National People's Congress Conference Center was bustling since early morning, with people discussing the varieties and prices involved in the negotiations.
It is reported that throughout today's negotiation process, representatives from companies such as Kelun Pharmaceutical, Otsuka Pharmaceutical (Japan), Fresenius Kabi, Baxter, Chia Tai Tianqing, Zai Lab, Simcere Pharmaceutical, Astellas, Nanjing Haina, and Beijing Ruiye entered the negotiation venue.
Referring to the previously released "List of Declared Drugs for Formal Review for the 2022 Adjustment of the National Reimbursement Drug List" by the National Healthcare Security Administration, the relevant varieties from the aforementioned companies mainly include Omadacycline Tosylate for Injection, Edaravone Dexborneol Concentrated Solution for Injection, Ceftazidime/Glucose (5%) for Injection, and Cefmetazole Sodium/Sodium Chloride for Injection. It is not difficult to predict that today's on-site negotiations will focus on areas such as respiratory and anti-infective drugs.
In 2021, a statement during the national medical insurance drug negotiations—"No group should be left behind"—resonated strongly with the public. Since the establishment of the National Healthcare Security Administration, the national drug list has been adjusted for four consecutive years, incorporating 507 new and effective drugs, removing 391 with unclear efficacy, and increasing the total number of drugs in the current list to 2,860. The average price reduction for drugs included in the list through negotiations exceeds 50%, effectively alleviating patient burdens.
Today, on the highly anticipated first day of the National Reimbursement Negotiations, what other updates are emerging?
"Generally Aligns with Company Expectations"
Based on historical negotiation data from 2018 to 2021, the average price reduction for drugs negotiated was approximately 50%-60%. Under the current simplified renewal rules, the price reduction for innovative drugs with new indications may be within 44%, which is narrower compared to the potential 50%-60% average reduction in renegotiations.
During the first day of negotiations, it was evident that companies with fewer varieties or a single product completed negotiations more efficiently, often within an hour. In contrast, companies with a larger number of varieties spent two to three hours or more from entry to exit.
The specific prices of the varieties negotiated on the first day have not yet been disclosed, but it is understood that multiple drugs involved today successfully completed negotiations. In interviews with reporters from the Pharmaceutical Economy News new media center, a company representative stated, "The negotiations generally met our expectations, but specific details cannot be disclosed."
Additionally, no oncology drugs were included in today's negotiations. Some company representatives who arrived early to "scout" the venue indicated that their oncology drug negotiations are scheduled for January 7.
In fact, prior to these negotiations, market expectations suggested that the price adjustments for this round of national drug list admission negotiations would be relatively moderate. Multiple securities firms' research reports noted that while maintaining the "basic coverage" principle, greater support is being given to "promoting innovation." Consequently, large pharmaceutical companies with significant innovation value, strong clinical efficacy, and cost advantages are expected to gain an edge in the intense negotiations and fully benefit from the medical insurance fund.
Some varieties potentially involved in today's National Reimbursement Negotiations
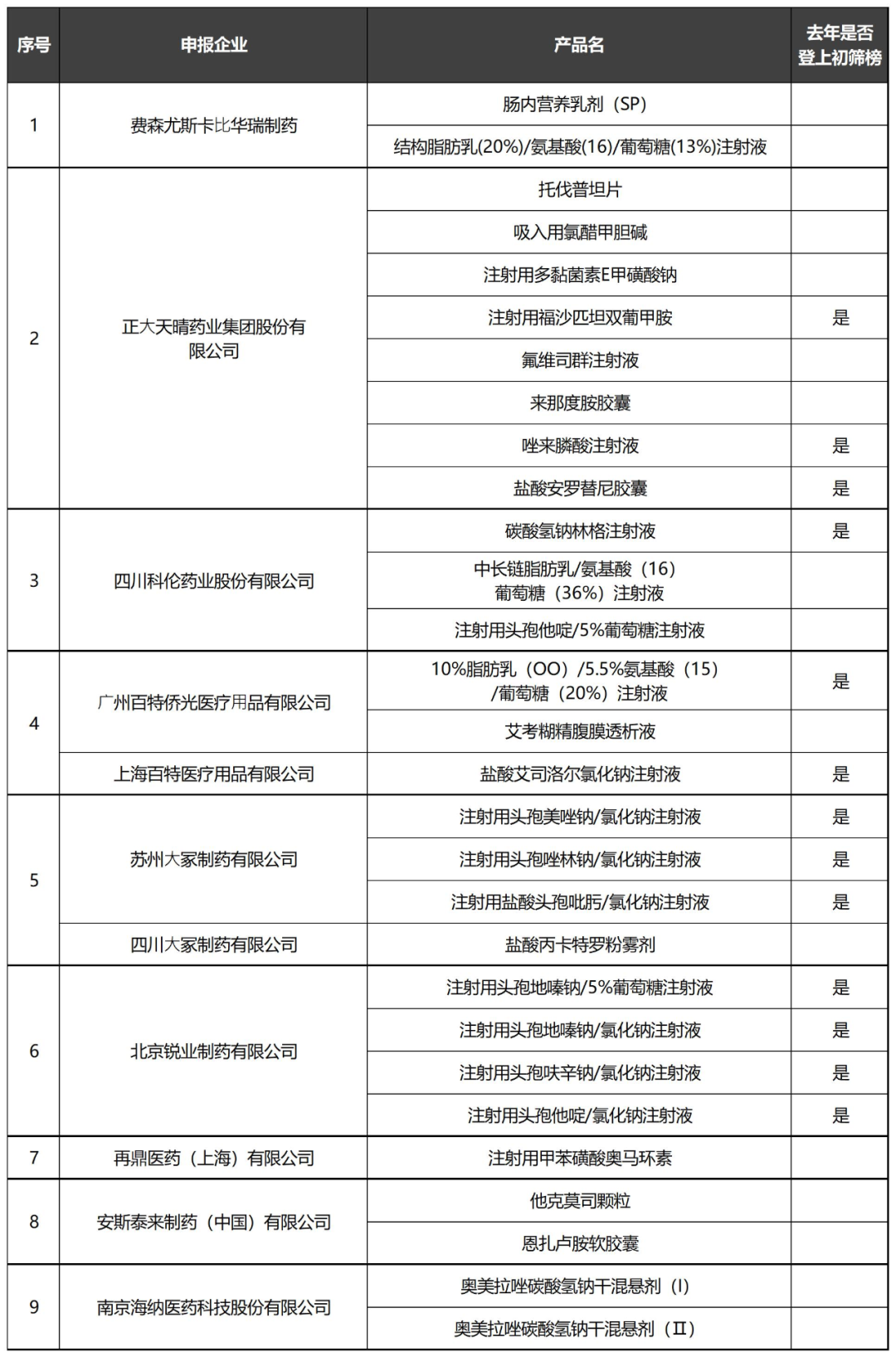
No New Information on CAR-T Products Yet
Relmacabtagene autoleucel injection, the second CAR-T drug in China and the sixth globally, developed and manufactured by JW Therapeutics, is undoubtedly the most anticipated highlight of this round of negotiations. This is the first time Relmacabtagene autoleucel injection has been shortlisted for the national negotiations. Its listed procurement price is RMB 1.29 million per injection, which is RMB 90,000 higher than Axicabtagene ciloleucel injection, which was not selected.
According to this year's negotiation rules for high-priced drugs undergoing formal review, the National Healthcare Security Administration stated that some relatively expensive drugs have passed the preliminary formal review, indicating only that they meet the application criteria and are eligible for the next stage. Whether such drugs can ultimately be included in the national drug list requires rigorous multi-faceted evaluations, including cost-effectiveness. Exclusive drugs must undergo negotiations, while non-exclusive drugs must go through a bidding process; only those successful in negotiations or bidding will be included.
Industry insiders suggest that given the current market and production status of CAR-T products, which involve personalized manufacturing, the production cost of Relmacabtagene autoleucel injection (excluding commercial costs) is considerable, potentially making it difficult to reduce the price to the medical insurance negotiation range. If JW Therapeutics does not demonstrate a clear intention to reduce the price, it is likely that the company will not participate in the national negotiations.
Non-Exclusive Drugs Draw Significant Attention
On June 13, 2022, the National Healthcare Security Administration released the "Work Plan for the 2022 Adjustment of the National Reimbursement Drug List for Basic Medical Insurance, Work-Related Injury Insurance, and Maternity Insurance," the "Renewal Rules for Negotiated Drugs," and the "Bidding Rules for Non-Exclusive Drugs" on its official website, marking the first public disclosure since the national drug admission negotiations began in 2016.
Following expert evaluation, non-exclusive drugs proposed for addition to the national drug list, excluding those selected in national centralized procurement or subject to government pricing, will be included through a bidding process.
The "Work Plan" stipulates that the medical insurance side will organize evaluation experts to conduct assessments, and companies participating in the application must submit bids according to the procedures. If at least one company participates and its bid does not exceed the medical insurance's willingness-to-pay, the generic drug will be included in Category B of the drug list; otherwise, it will not be included. For drugs included via bidding, the lowest bid among all companies will serve as the reimbursement standard for that generic drug.
These rules indicate that after the new list is released, non-exclusive drugs entering the list will have a unified medical insurance reimbursement standard, establishing a new method for determining such standards.
The "List of Declared Drugs for Formal Review for the 2022 Adjustment of the National Reimbursement Drug List for Basic Medical Insurance, Work-Related Injury Insurance, and Maternity Insurance" shows 198 new non-reimbursed products, of which 41 are non-exclusive products—40 are Western medicines and 1 is a proprietary Chinese medicine.
Non-exclusive products have consistently been key players in price reductions. To secure inclusion in the medical insurance list in 2022, companies must also navigate the new approach of price bidding for reimbursement.
Notably, under the rule that the lowest bid determines the reimbursement standard, drugs selected through bidding will passively enter the list at low prices. Products from companies that bid higher or did not participate will also be reimbursed at this price. Some industry experts predict that "price competition may be as intense as in centralized procurement." Others suggest that this rule primarily targets high-priced products sold mainly outside hospital settings, serving as a complementary and exploratory measure to the centralized procurement policy.
Taking Memantine Hydrochloride Extended-Release Capsules, one of the most competitively contested varieties, as an example, five companies applied: Zhejiang Jingxin Pharmaceutical, Qingdao Baoyang Pharmaceutical, Jiangsu Tasly Diyi Pharmaceutical, Chengdu Easton Biopharmaceutical, and Fujian Baonuo Pharmaceutical. Additionally, Fosaprepitant Dimeglumine for Injection is also contested by multiple companies, including Shandong Luoxin, Jiangsu Hansoh, and Chia Tai Tianqing.
From a therapeutic area perspective, in the upcoming rounds of medical insurance negotiations, fields such as neurology, endocrinology and metabolism, cardiovascular diseases, and oncology remain under close market scrutiny.