



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2022-09-16
Posting Date:2022-09-16 Views:
Views: Exciting Content
To meet the needs of clinical medication and standardized drug management, since 2004, the National Medical Products Administration (NMPA) has converted 805 prescription drugs (Rx) into over-the-counter (OTC) drugs. These include many Category A OTC and dual-category varieties such as Omeprazole Enteric-coated Tablets, Pudilan Anti-inflammatory Tablets, and Budesonide Nasal Spray. Data from Menet shows that in 2021, the sales scale of the China retail pharmacy terminal (urban实体 pharmacies + online pharmacies) exceeded RMB 300 billion, a year-on-year increase of 11.42%. With an increasing number of prescription drugs being converted to OTC, the retail pharmacy market is poised for a significant reshuffle.
46 Prescription Drugs Converted to OTC, 15 Category A and Dual-Category Varieties Included
Drugs can be classified as prescription (Rx) drugs and over-the-counter (OTC) drugs. However, the classification of a drug is not static; under certain conditions, a prescription drug can be converted to an OTC drug. Among OTC drugs, Category A OTC drugs must be purchased and used under the guidance of a licensed pharmacist in a pharmacy, while Category B OTC drugs do not have this requirement. Typically, a drug is classified either as a prescription drug or an OTC drug. However, sometimes a drug can be both a prescription drug and an OTC drug, which is also known as a dual-category variety.
Since 2021, the NMPA has issued 21 announcements regarding the conversion of prescription drugs to OTC drugs, involving 46 types of drugs (by product name). These include Omeprazole Enteric-coated Tablets, Glucosamine Potassium Sulfate Tablets, Baofukang Gel, Tiaojing Yangyan Granules, Pudilan Anti-inflammatory Tablets, Honghua Xiaoyao Tablets, Felbinac Gel, Aloe Pearl Capsules, Buxu Xiaoke Mixture, Ambroxol Hydrochloride Sustained-release Tablets, Shufeng Jiedu Capsules, Jinzhen Granules, and Fuke Baifeng Capsules, among others. According to relevant requirements, these drugs can be sold through retail channels without a prescription.
List of Prescription Drugs Converted to OTC Published by NMPA in the Past Two Years
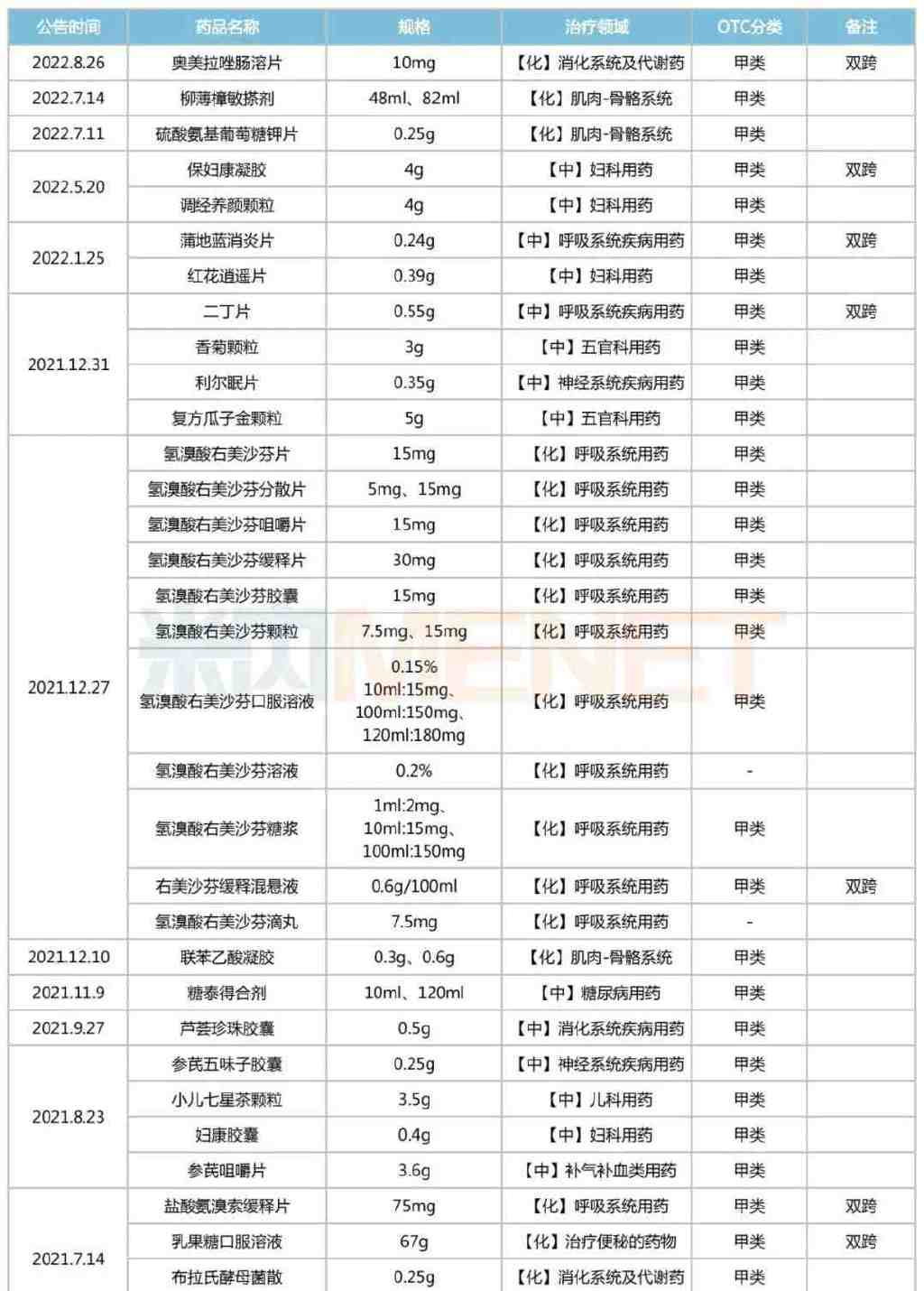
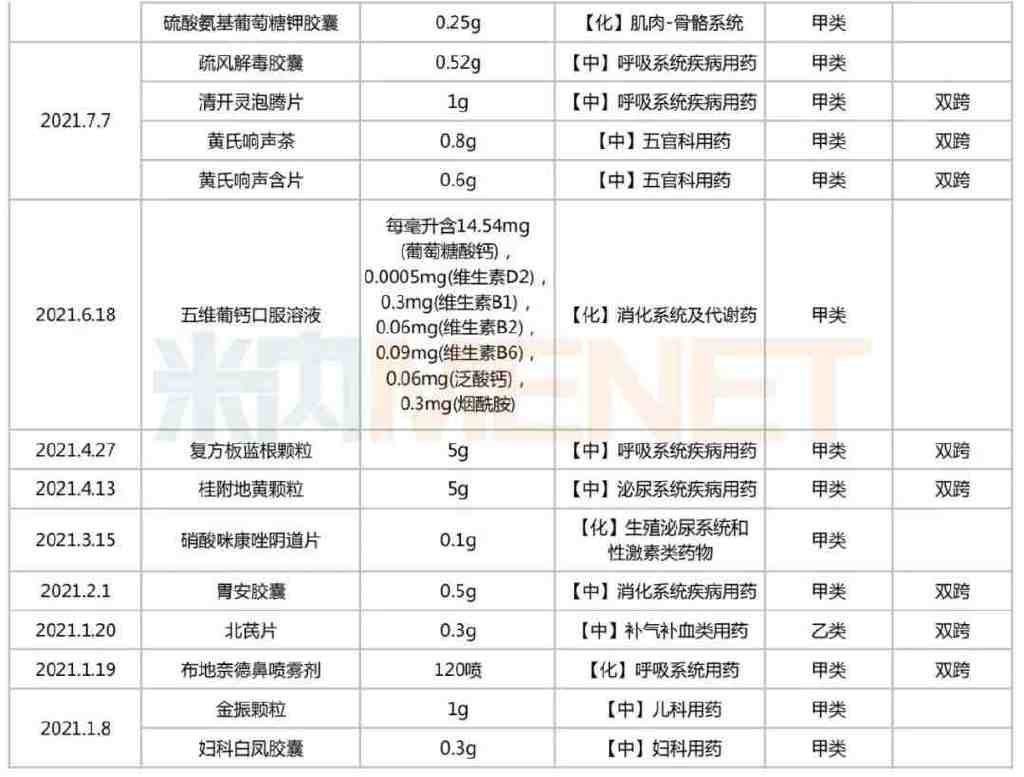
Source: National Medical Products Administration (NMPA), compiled by Menet
Among the 46 prescription drugs converted to OTC, there are 15 dual-category varieties. These include commonly used clinical drugs such as Omeprazole Enteric-coated Tablets, Baofukang Gel, Pudilan Anti-inflammatory Tablets, Ambroxol Hydrochloride Sustained-release Tablets, Lactulose Oral Solution, Compound Banlangen Granules, and Budesonide Nasal Spray. In terms of therapeutic category, chemical drugs for the respiratory system are the most numerous, accounting for 13 varieties. They are followed by Chinese patent medicines for the respiratory system, chemical drugs for the musculoskeletal system, Chinese patent medicines for gynecological use, and Chinese patent medicines for ENT and ophthalmology, each with 4 or more varieties.
Pudilan Anti-inflammatory, Budesonide... in Full Swing, Stirring Up the RMB 300 Billion Retail Pharmacy Market Again
In the past two years, multiple prescription drugs converted to OTC have seen their sales and growth rates continue to rise in the retail pharmacy terminal.
Pudilan Anti-inflammatory Tablets, known for their "clearing heat and detoxifying" effects, had only tens of millions of yuan in sales revenue in China's public medical institution terminal (urban public hospitals, county-level public hospitals, urban community centers, and township卫生院) in 2021. However, their sales scale in the China retail pharmacy terminal approached RMB 800 million, a year-on-year increase of 9.42%. This indicates that for such products used to treat common colds and coughs rather than antibiotics, the retail market is clearly more favorable than the hospital market.
Sales trend of Pudilan Anti-inflammatory Tablets in China's retail pharmacy terminal in recent three years (Unit: RMB 10,000)
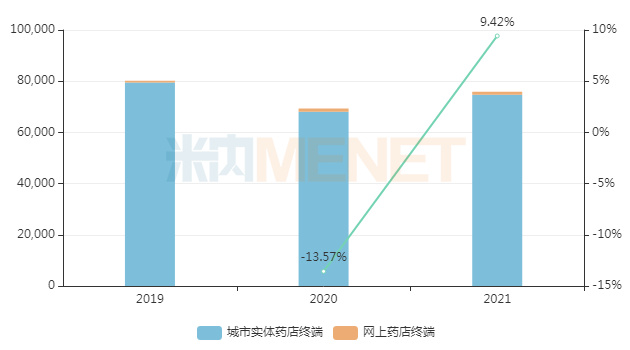
Source: Menet Database
Budesonide is a major inhaled drug, and Budesonide Nasal Spray is its second best-selling formulation. In 2021, its sales scale in China's public medical institution terminal exceeded RMB 400 million, while its sales in the China retail pharmacy terminal also exceeded RMB 200 million. Among these, the sales growth rate in the online pharmacy terminal was as high as 63.21%. Currently, only two import companies, AstraZeneca and Chiesi Pharmaceuticals, are approved to produce this drug. For exclusive products or those with a less competitive landscape undergoing an "Rx-to-OTC switch," the retail pharmacy terminal is expected to become a significant driver of sales growth.
Approval Status of Budesonide Nasal Spray Manufacturers
Source: Menet New Version Database
Omeprazole Enteric-coated Tablets are a proton pump inhibitor (PPI) mainly used to treat duodenal ulcers, Zollinger-Ellison syndrome (gastrinoma), gastric ulcers, and reflux esophagitis. Affected by centralized procurement, the sales of this drug in China's public medical institution terminal dropped by 76.89% in 2021. In contrast, in the China retail market, especially the online pharmacy terminal, its sales maintained a year-on-year growth rate of 53.17%, indicating remaining potential. As a major variety in centralized procurement, its profits in the hospital market have been significantly compressed. Now that it has been converted from a prescription drug to an OTC drug, relevant pharmaceutical companies are expected to seize the opportunity to accelerate their expansion into the retail market, filling the profit gap caused by the impact of centralized procurement.
Sales trend of Omeprazole Enteric-coated Tablets in China's online pharmacy terminal in recent three years (Unit: RMB 10,000)
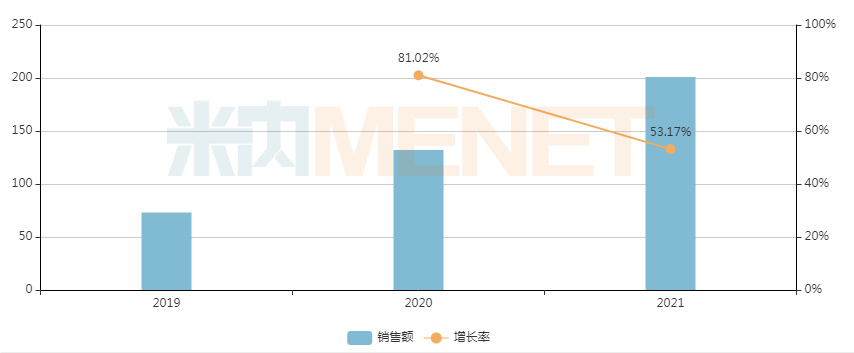
Source: Menet China Online Pharmacy Terminal Competitive Landscape Database
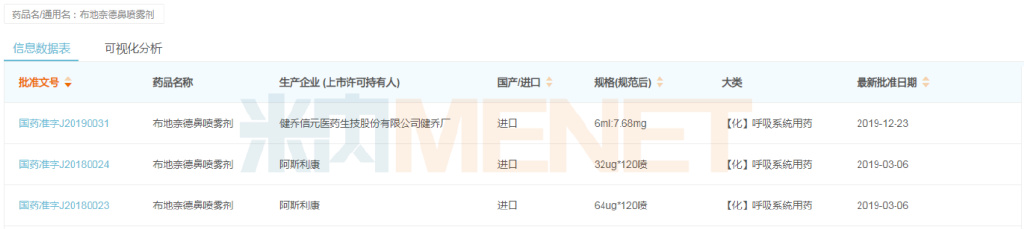
Menet data shows that in 2021, the total market size for chemical drugs and Chinese patent medicines in China's retail pharmacy terminal exceeded RMB 300 billion, a year-on-year increase of 11.42%. According to不完全统计, since 2004, a total of 805 drugs have been converted from prescription drugs to OTC drugs, involving thousands of manufacturers. As more and more prescription drugs are converted to OTC, the retail market is set to face a new wave of intense competition.
Overall Sales Trend of Chemical Drugs and Chinese Patent Medicines in China's Retail Pharmacy Terminal, 2019-2021 (Unit: RMB 100 million)
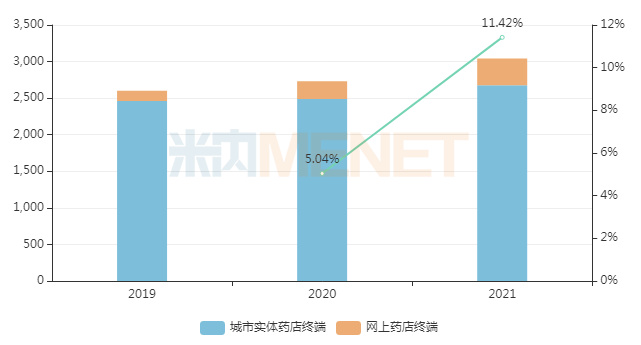
Source: Menet Database
"Rx-to-OTC Switch" or Becoming a Major Trend: What is the Intention Behind It?
The expansion of the "Rx-to-OTC switch" is supported by the continuous improvement of China's drug management system. With the implementation of relevant policies, a situation is emerging where multiple parties benefit.
1. Benefits for medication consumers
According to the classified management requirements for prescription drugs and Category A/Category B OTC drugs under the "Drug Administration Law," when a drug is converted from a prescription drug to an OTC drug, patients can purchase it from offline pharmacies and online e-commerce platforms without a doctor's prescription. This increases purchase channels and makes drug acquisition more convenient, which is particularly beneficial for patients who require long-term medication.
2. Reduces national medical insurance expenditure
Expenditure on medical care and pharmaceuticals increases the government's fiscal burden. Varieties converted from prescription drugs to Category B OTC drugs will exit the national medical insurance reimbursement system. In the long run, "Rx-to-OTC switch" will be an effective measure to save national medical insurance funds.
3. Saves clinical diagnostic and treatment resources
For some minor ailments, patients can achieve recovery through self-diagnosis and self-medication. This eliminates the need for doctors to spend time and effort on prescribing, allowing them more time to treat patients with serious illnesses, improve their diagnostic and treatment skills, and gain self-satisfaction and a sense of accomplishment from their work and patient satisfaction.
4. Expands retail channels and extends product lifecycle
Drugs, like other products, have a lifecycle. After the patent for an original drug expires, it faces intense price competition from generic drugs, leading to a sharp decline in sales—a phenomenon known as the patent cliff. This is compounded by recent policy impacts such as medical insurance cost control and price reductions through centralized procurement, which have even forced some original drugs out of the market. By converting a prescription drug to an OTC drug, on one hand, the product's lifecycle can be extended, allowing the pharmaceutical company to gain more revenue. On the other hand, the target audience for the product expands from specific patients to the general public, which can help drive sales of the company's other products.
5. Enriches product variety and drives new growth in the retail industry
For terminal pharmacies, a greater variety of OTC drugs better meets consumer needs. "Rx-to-OTC switch" drugs can be purchased directly in pharmacies, potentially increasing product sales volume and expanding market share. If a drug already has a strong consumer base in its prescription drug stage, opening up new channels after the "Rx-to-OTC switch" can bring even greater profits to the company. In recent years, China's OTC market has grown rapidly. "Rx-to-OTC switch" varieties gain access to more flexible promotion and sales channels, presenting significant development opportunities for the OTC market and related companies.
Conclusion
It is foreseeable that due to factors such as volume-based procurement, medical insurance cost control, and healthcare reforms, a large number of prescription drugs will be squeezed out of hospital systems. Industry insiders speculate that in the future, about 70% of routine prescription drugs will be converted to non-prescription drugs (general medicines), and these drugs will gradually move to the out-of-hospital market, unleashing significant market potential. "How to leverage the policy windfall of the Rx-to-OTC switch to quickly bring the advantages of prescription drugs into the OTC market, delivering products with stronger professionalism, clearer efficacy, and better consumer perception" will become a key competitive focus for companies in the next phase.