



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2022-08-26
Posting Date:2022-08-26 Views:
Views: Recently, a notice regarding procurement data related to the scope of varieties for the first batch of continuation and the third batch of centralized drug procurement in Zhejiang's public medical institutions (hereinafter referred to as the "Volume Reporting Notice") has been circulating within the industry. Twenty chemical drugs, including lansoprazole, along with 2 varieties not renewed from the first batch (amoxicillin clavulanate potassium, ceftizoxime), appear in the formal volume reporting variety list. The total sales scale of these 22 varieties in Zhejiang's public hospital terminal in 2021 was close to RMB 400 million. Based on the release time of the "Volume Reporting Notice," it is speculated that the bid opening for the third batch of centralized procurement may be moved forward.
The "lowest bidder wins" scenario is no longer the case, highlighting the advantages of the "dual envelope" model
Since 2020, provincial/provincial alliance centralized procurement has flourished across the country. Various regions have successively carried out special centralized procurements for injections, insulin, antibacterial drugs, etc., based on their actual circumstances, with procurement rules and selection models varying.
In Zhejiang Province, the innovative aspect lies in dividing the varieties included in volume-based procurement into Group A (originator drugs or reference-listed drugs, generic drugs that have passed consistency evaluation) and Group B (other generic drugs), i.e., the "dual envelope" model. If either Group A or Group B fails to have a selected drug, 50% of the agreed procurement volume of that group will be merged into the agreed procurement volume of the other group, and the remaining 50% will be treated as residual usage outside the agreed procurement volume. This is a positive signal for both selected and non-selected varieties.
● Regarding Agreed Procurement Volume – Calculated as 60% of the total annual usage of drugs by all public medical institutions in the province in 2021, divided into three scenarios:
(1) Both groups have selected drugs: The agreed procurement volumes for Groups A and B are determined based on their respective procurement shares from all public medical institutions in the province in 2021 (the maximum and minimum agreed procurement volumes are 50% and 10% of the total annual usage of the variety, respectively);
(2) No selected drug in either Group A or Group B: 50% of the agreed procurement volume of the group without a selected drug is merged into the other group, and the remaining 50% is treated as residual usage outside the agreed procurement volume;
(3) Only 50% of the original agreed procurement volume is obtained by either Group A or Group B: Half of the other 50% of the original agreed procurement volume of that group is merged into the other group, and the remaining half is treated as residual usage outside the agreed procurement volume.
● Regarding Procurement Rules – The technical evaluation part for the third batch is roughly the same as the second batch, with a total of 6 indicators, including clinical efficacy, quality reliability, clinical safety, brand recognition, packaging quality and convenience, and service reputation.
Technical Evaluation Scoring Table
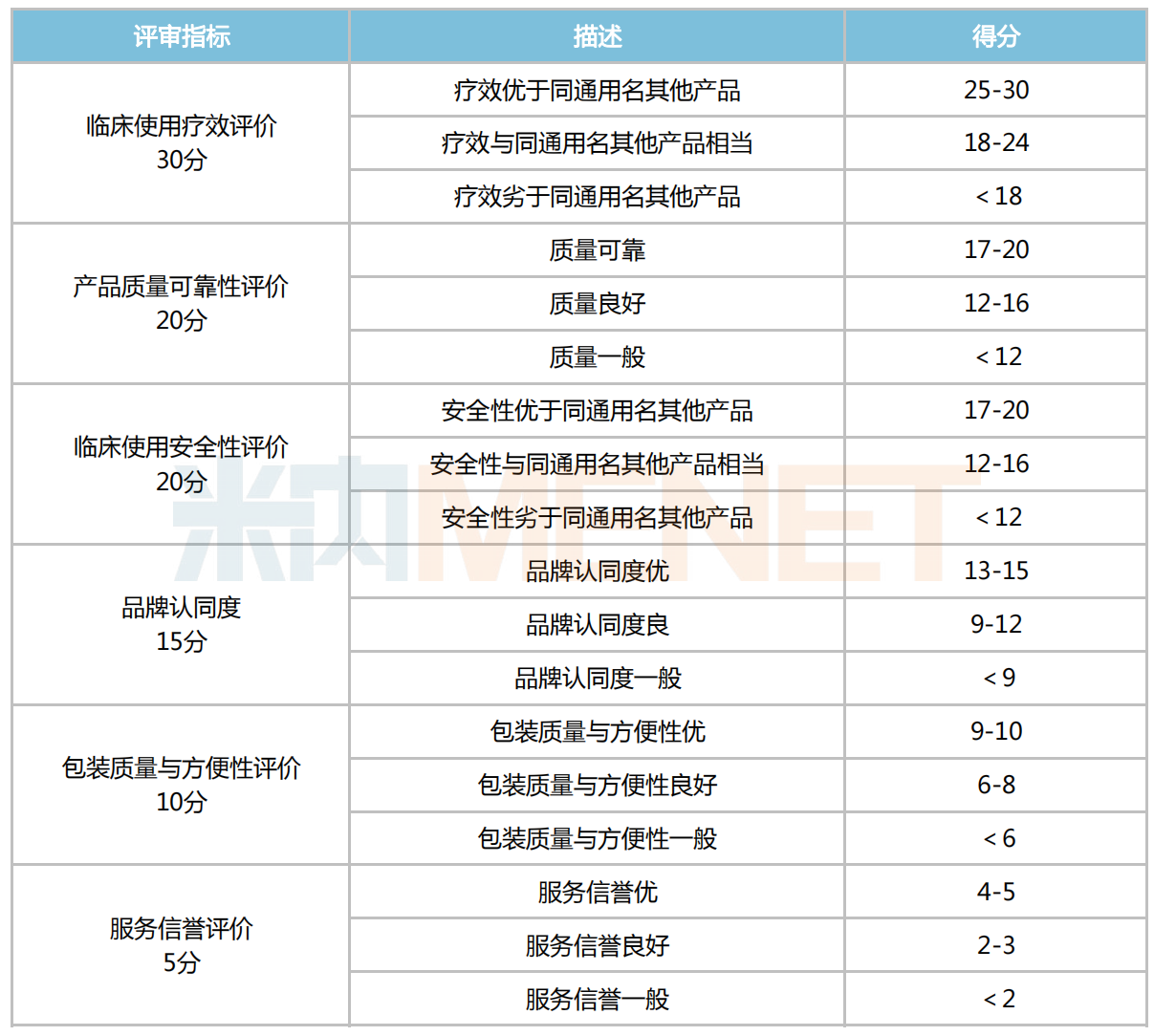
Based on the expert scores, the highest and lowest scores are removed, and the average score (rounded to two decimal places) is taken as the expert evaluation result. Groups A and B for each variety are ranked from highest to lowest score. If there is a tie for the last qualifying position, all tied companies proceed to the price evaluation stage.
Companies Entering Price Evaluation
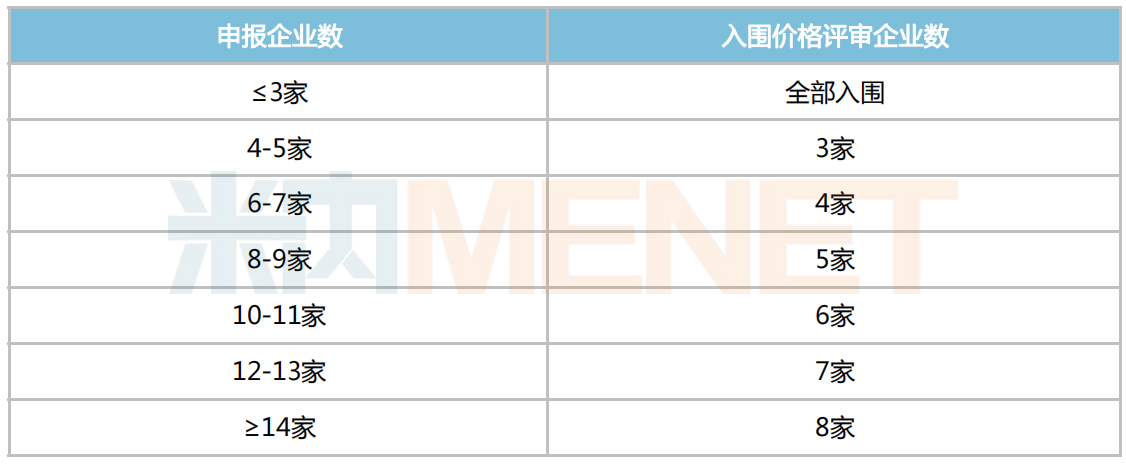
The price evaluation part is also largely similar to the second batch of centralized procurement, divided into two scenarios: number of applying companies = 1, and number of applying companies ≥ 2. The specific details for proposed selection are shown in the figure below:
Proposed Selection Details for Price Evaluation
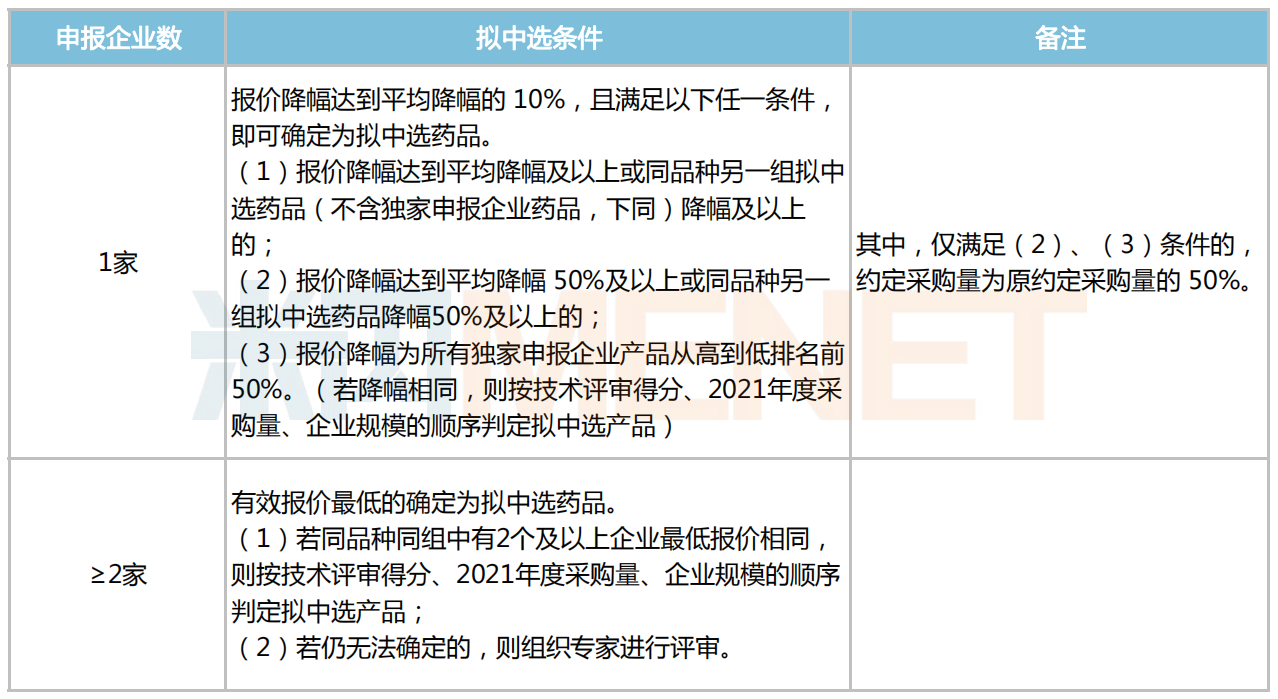
The third batch of centralized procurement in Zhejiang will continue to use the "dual envelope" model combining technical evaluation and price evaluation. As can be seen from the technical evaluation indicators, non-price factors such as efficacy, quality, and safety account for a relatively high proportion of the score, reflecting the simultaneous requirements for drug quality and price. Under the "dual envelope" model, companies must not only control product prices but also pay more attention to cultivating their "internal strengths" such as product advantages and solid technical indicators.
Additionally, it is worth noting that the "Volume Reporting Notice" requires medical institutions to report volumes scientifically and accurately based on factors such as changes in medication structure and adjustments to medical insurance payment policies. If the reported volume shows an increase of 100% or a decrease of 20% compared to the actual procurement (distribution) volume, the system will issue a warning prompt.
RMB 400 Million Market Reshuffled! Rabeprazole, Cefotaxime... Product Landscape Reshaped
Annual provincial-level volume-based procurement has become normalized across provinces. Looking back at the first two batches of drug volume-based procurement in Zhejiang Province, the first batch planned to include 8 varieties with 16 specifications, with 10 drugs ultimately selected; the second batch planned to include 17 varieties with 34 specifications, with 21 drugs ultimately selected.
Selected Drug List for Zhejiang's First Batch of Centralized Procurement
Selected Drug List for Zhejiang's Second Batch of Centralized Procurement
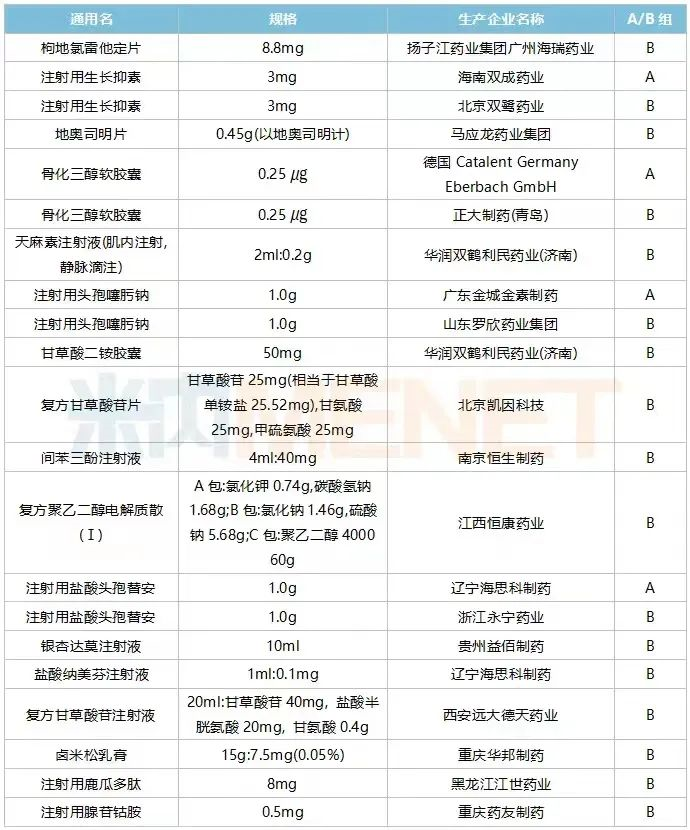
Source: Official Website of Zhejiang Pharmaceutical Procurement Center
As a product from the first batch of centralized procurement, Rabeprazole Sodium Enteric-coated Tablets saw a 45.18% increase in sales at public hospitals in Zhejiang Province in 2021. Two companies were selected. Eisai (originator) remained strong, maintaining a market share of over 90% since 2021. Double-Crane Pharmaceutical (Hainan) experienced steady growth, with a market share of 2% to 3% in the past two years, ranking third in the brand pattern for this product.
Ceftizoxime Sodium for Injection saw a 70.27% increase in sales at public hospitals in Zhejiang Province in 2021. Leveraging the advantage of being selected in centralized procurement, Guangdong Golden Kangjin and Shandong Luoxin both achieved a "breakthrough from zero" in market share in Q1 2022, together accounting for over 16% of the market.
Another popular product, Desloratadine Citrate Disodium Tablets, saw a 34.31% increase in sales at public hospitals in Zhejiang Province in 2021. This product is exclusively manufactured by Yangtze River Pharmaceutical Group Guangzhou Hairui Pharmaceutical. Based on Q1 2022 data, it is projected that the annual sales of this product may reach a new historical high.
Haisco Pharmaceutical had two products selected in the second batch of centralized procurement. Among them, Nalmefene Hydrochloride Injection saw an 8.25% increase in sales at public hospitals in Zhejiang Province in 2021. Haisco's market share for this product was less than 8%, but being selected is expected to help reverse this trend.
Sales Scale in Zhejiang Public Hospital Terminal in Recent Years (Unit: RMB 100 million)
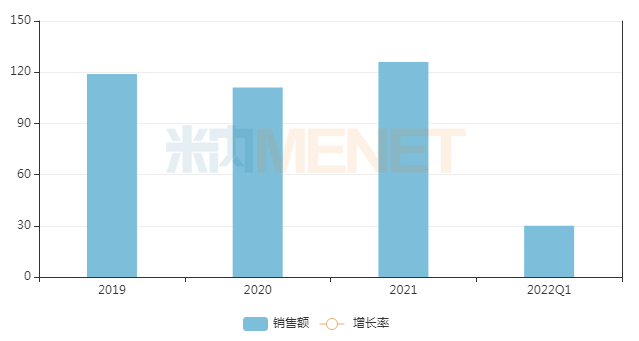
Source: Menet Key Provincial/City Public Hospital Terminal Competitive Landscape Database
Menet data shows that sales in Zhejiang Province at the key provincial/city public hospital terminal exceeded RMB 10 billion in both 2020 and 2021. To date, the two batches of Zhejiang provincial centralized procurement have included 31 drugs, with total sales of approximately RMB 400 million in the province's public hospital terminal in 2021.
The Third Batch of Centralized Procurement is on the Way: 22 Varieties, Including Lansoprazole and Leuprorelin, to be Included
Looking at the timeline of drug centralized procurement in Zhejiang Province in recent years, the first two batches both had draft opinions released in October, proposed selected variety lists announced in December, and final selection results announced in January of the following year. The "Volume Reporting Notice" for this third batch was released in late August, suggesting that the bid opening date for this batch may be moved forward. This move might be to allow sufficient preparation time for the eighth round of national centralized procurement and the 2022 national reimbursement negotiations.
According to the "Volume Reporting Notice," 22 chemical drugs (including varieties not renewed from the first batch, the same below), such as lansoprazole and leuprorelin, are planned to be included in the third batch of centralized procurement in Zhejiang Province. The 10 biologics, including growth hormone, which were previously rumored, have not yet appeared in the volume reporting list for this batch.
List of Varieties for Zhejiang's Third Batch of Centralized Procurement
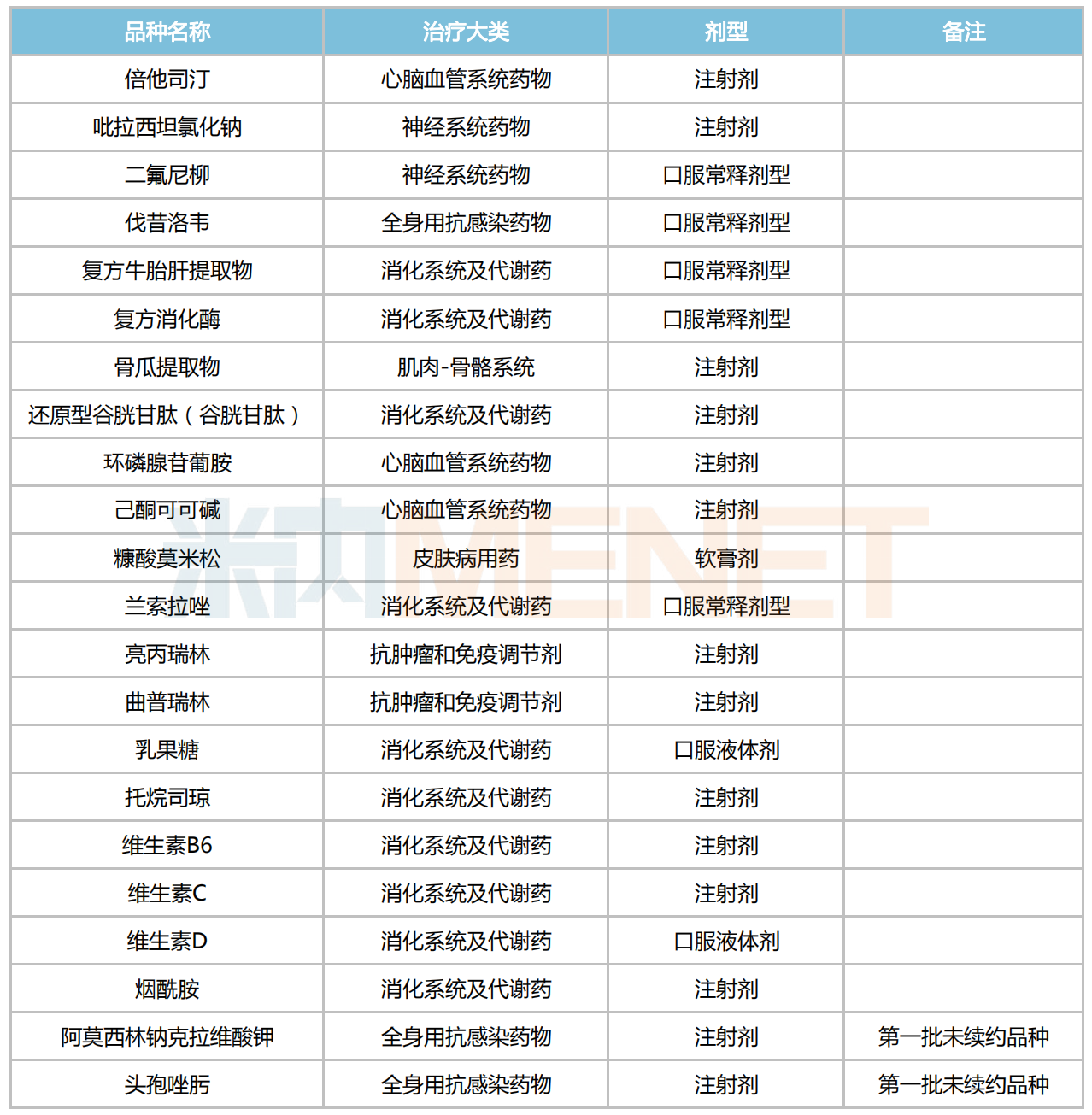
Menet data shows that the total sales scale of these 22 varieties in Zhejiang's public hospital terminal in 2021 was close to RMB 400 million. By therapeutic category, drugs for the digestive system and metabolism accounted for 10 spots, drugs for the cardiovascular system and systemic anti-infectives each accounted for 3 spots, and anti-tumor and immunomodulators and nervous system drugs each accounted for 2 spots.
Status of Generic Consistency Evaluation for Varieties Planned for the Third Batch of Centralized Procurement
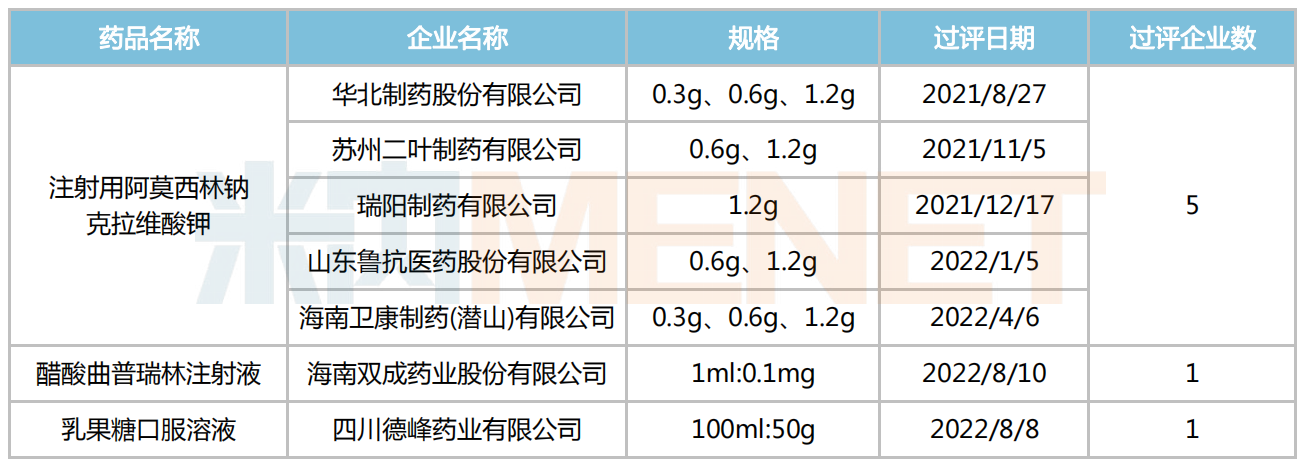
Note: Varieties not listed indicate no company has passed the consistency evaluation yet.
Source: Menet MED2.0 China Drug Review Database
From the perspective of consistency evaluation status, except for Amoxicillin Sodium Clavulanate Potassium for Injection, Triptorelin Acetate Injection, and Lactulose Oral Solution, most of the varieties planned for inclusion in this batch have not yet had any company pass the consistency evaluation.
In fact, provincial/provincial alliance centralized procurement functions more like a supplement after national centralized procurement, a "relay baton" for market resource reallocation. Implementing and advancing provincial/provincial alliance centralized procurement reflects the guiding principle proposed by the state: "Adhere to volume-based procurement, ensure quality, promote innovation-driven development, ensure supply stability, ensure clinical use, and set bidding rules based on specific varieties and competitive landscape."