



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-06-02
Posting Date:2023-06-02 Views:
Views: To meet the needs of clinical use and standardized drug management, the NMPA has switched 813 prescription drugs to over-the-counter (OTC) drugs since 2004. These include commonly used drugs such as Pudilan Xiaoyan Tablets, Budesonide Nasal Spray, and Lactulose Oral Solution. Data from Minenet shows that the total sales of chemical drugs and TCM patent medicines in China’s retail pharmacy terminal (urban physical pharmacies + online pharmacies) approached RMB 350 billion in 2022, representing a year-on-year increase of 13.02%. As more and more prescription drugs are switched to OTC drugs, the retail pharmacy market is poised for a major reshuffle.
54 Prescription Drugs Switched to OTC | TCM Patent Medicines Dominate, Exclusive Products Stand Out
Drugs can be classified as prescription (Rx) drugs or over-the-counter (OTC) drugs. However, the classification of a drug is not static; under certain conditions, a prescription drug can be switched to an OTC drug. Among these, Category A OTC drugs must be purchased and used under the guidance of a licensed pharmacist in a pharmacy, while Category B OTC drugs do not have this requirement. Typically, a drug is classified as either a prescription drug or an OTC drug, but sometimes a drug can be both, also known as a dual-status drug.
Since 2021, the NMPA has issued 25 announcements regarding the switch of prescription drugs to OTC drugs, covering a total of 54 drugs (counted by product name). These include Ambroxol Hydrochloride Orally Disintegrating Tablets, Ejiao Capsules, Codonopsis Oral Liquid, Omeprazole Enteric-coated Tablets, Pudilan Xiaoyan Tablets, Xiao’er Qixingcha Granules, Budesonide Nasal Spray, Jinzhen Granules, and others. According to relevant requirements, these drugs can be sold through retail channels without a prescription.
List of Prescription Drugs Switched to OTC Announced by NMPA Since 2021
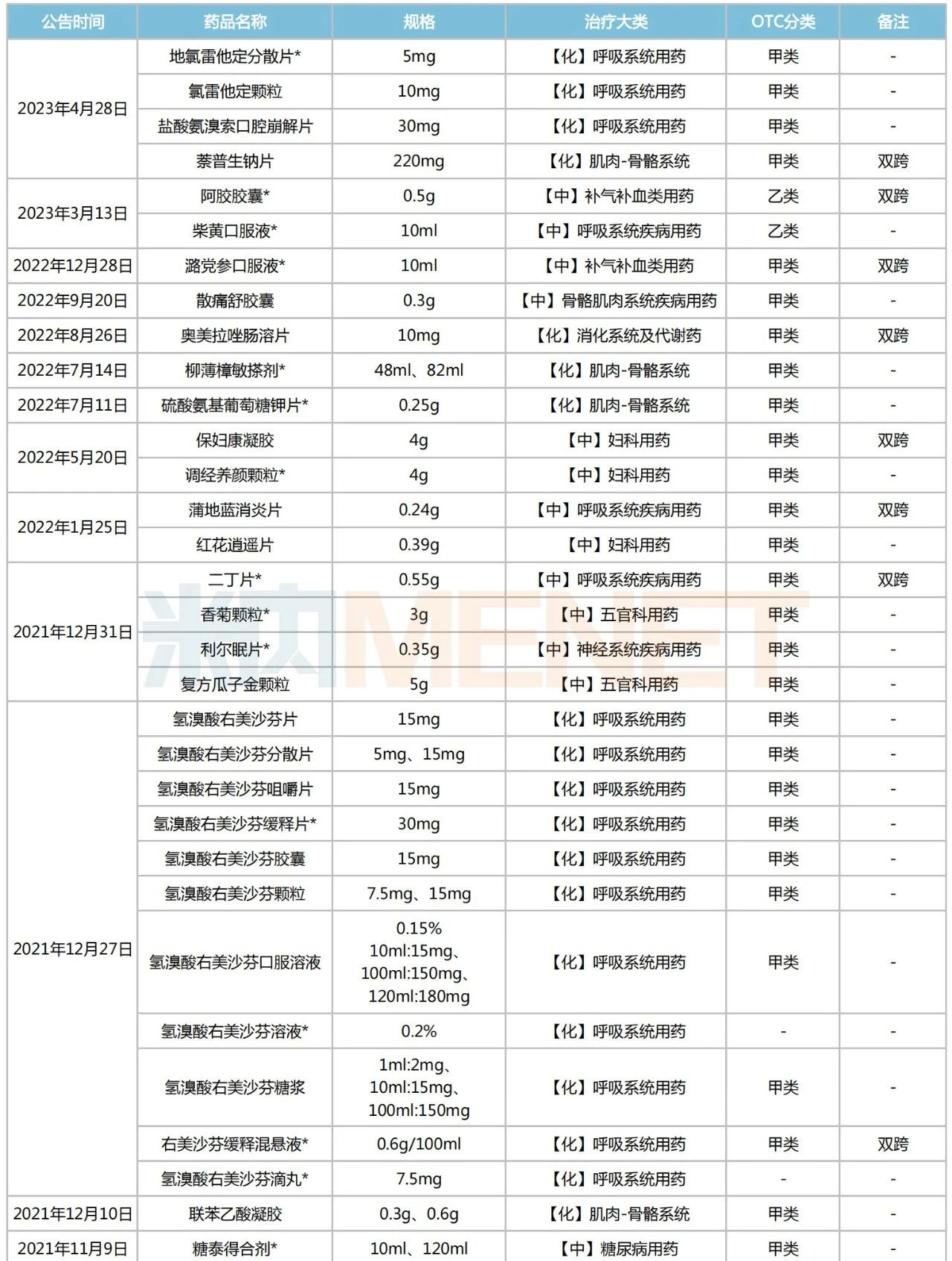
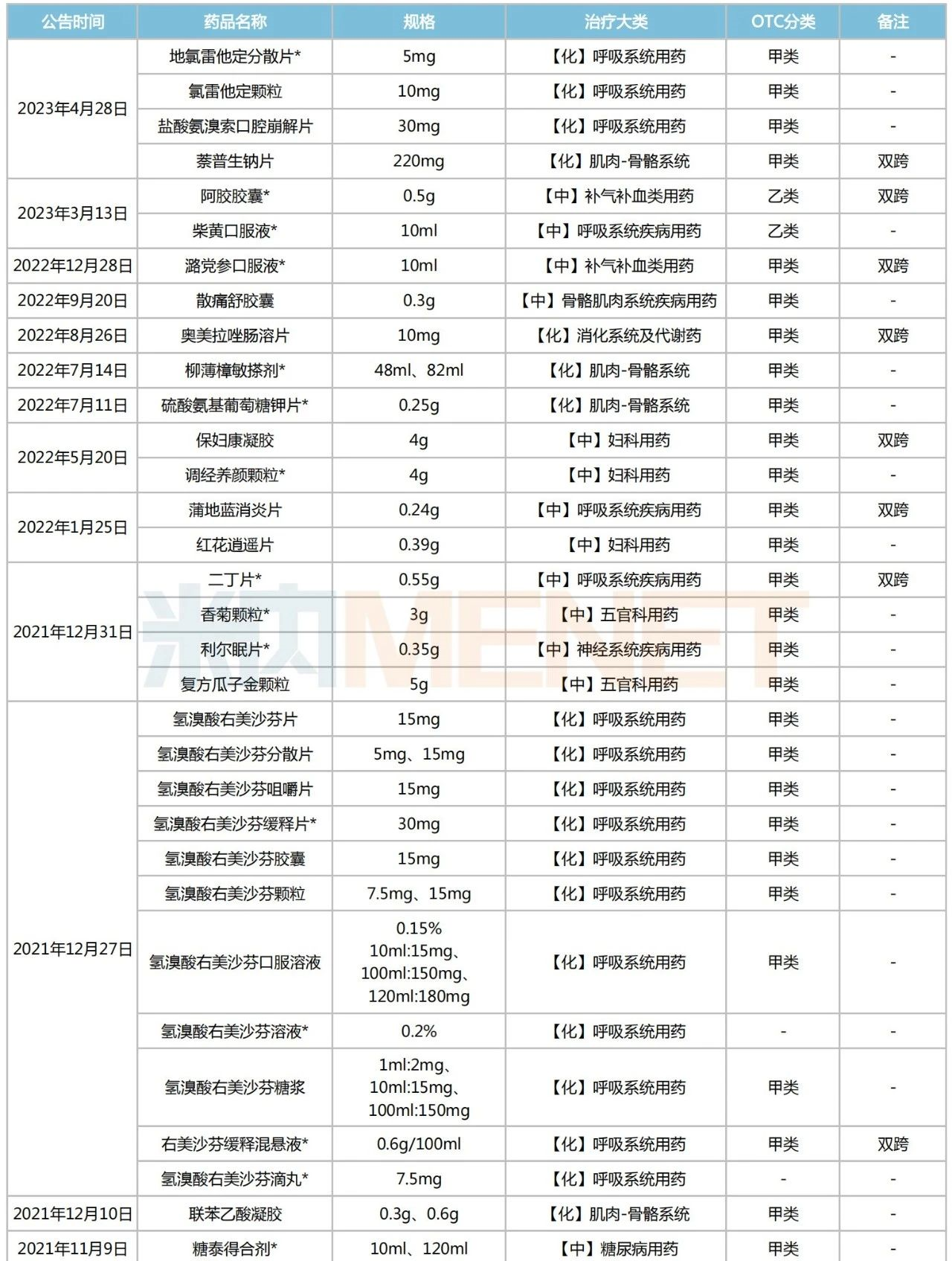
Note: * indicates exclusive varieties (including exclusive dosage forms)
Among the 54 prescription drugs switched to OTC, 26 are exclusive varieties. TCM patent medicines account for over 60%, with 16 varieties. There are 18 dual-status varieties, including commonly used clinical drugs such as Pudilan Xiaoyan Tablets, Codonopsis Oral Liquid, Omeprazole Enteric-coated Tablets, Ambroxol Hydrochloride Sustained-release Tablets, Lactulose Oral Solution, and Budesonide Nasal Spray. By therapeutic category, chemical drugs for the respiratory system are the mainstay, totaling 16 varieties. These are followed by TCM patent medicines for respiratory system diseases (6 varieties), chemical drugs for the musculoskeletal system (5 varieties), and TCM patent medicines for gynecological use (5 varieties).
Pudilan Xiaoyan, Budesonide… Promising Potential, Stirring Up the RMB 350 Billion Market
In the past three years, the sales and growth rates of several prescription drugs switched to OTC have continued to rise in the retail pharmacy terminal.
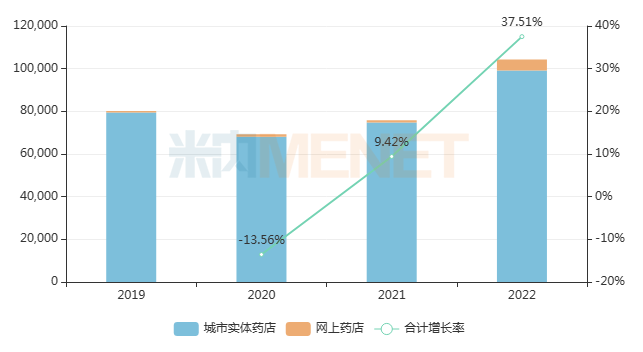
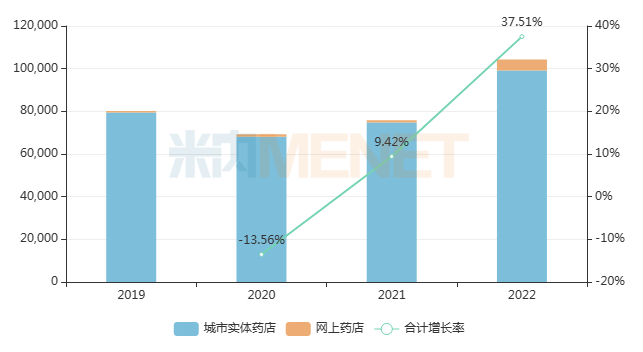
Pudilan Xiaoyan Tablets, known for their “clearing heat and detoxifying” effect, recorded only tens of millions of yuan in sales in China’s public medical institutions (urban public hospitals, county-level public hospitals, community health centers, and township health centers) in 2022. However, their sales in the retail pharmacy terminal exceeded RMB 1 billion, with a year-on-year increase of 37.51%. It is evident that such products for common colds and coughs, rather than antibiotics, perform significantly better in the retail market than in the hospital market.
Recent Sales Trends of Pudilan Xiaoyan Tablets in China’s Retail Pharmacy Terminal (Unit: RMB 10,000)
Lactulose Oral Solution is a commonly used constipation treatment for children in clinical practice. Over a dozen domestic manufacturers are approved to produce it. According to Minenet data, sales of this product across the three major terminals and six major markets in China have been increasing year by year, exceeding RMB 2.1 billion in 2022, with a year-on-year growth of 14.45%. Among chemical drugs for constipation, Lactulose Oral Solution consistently ranks first.
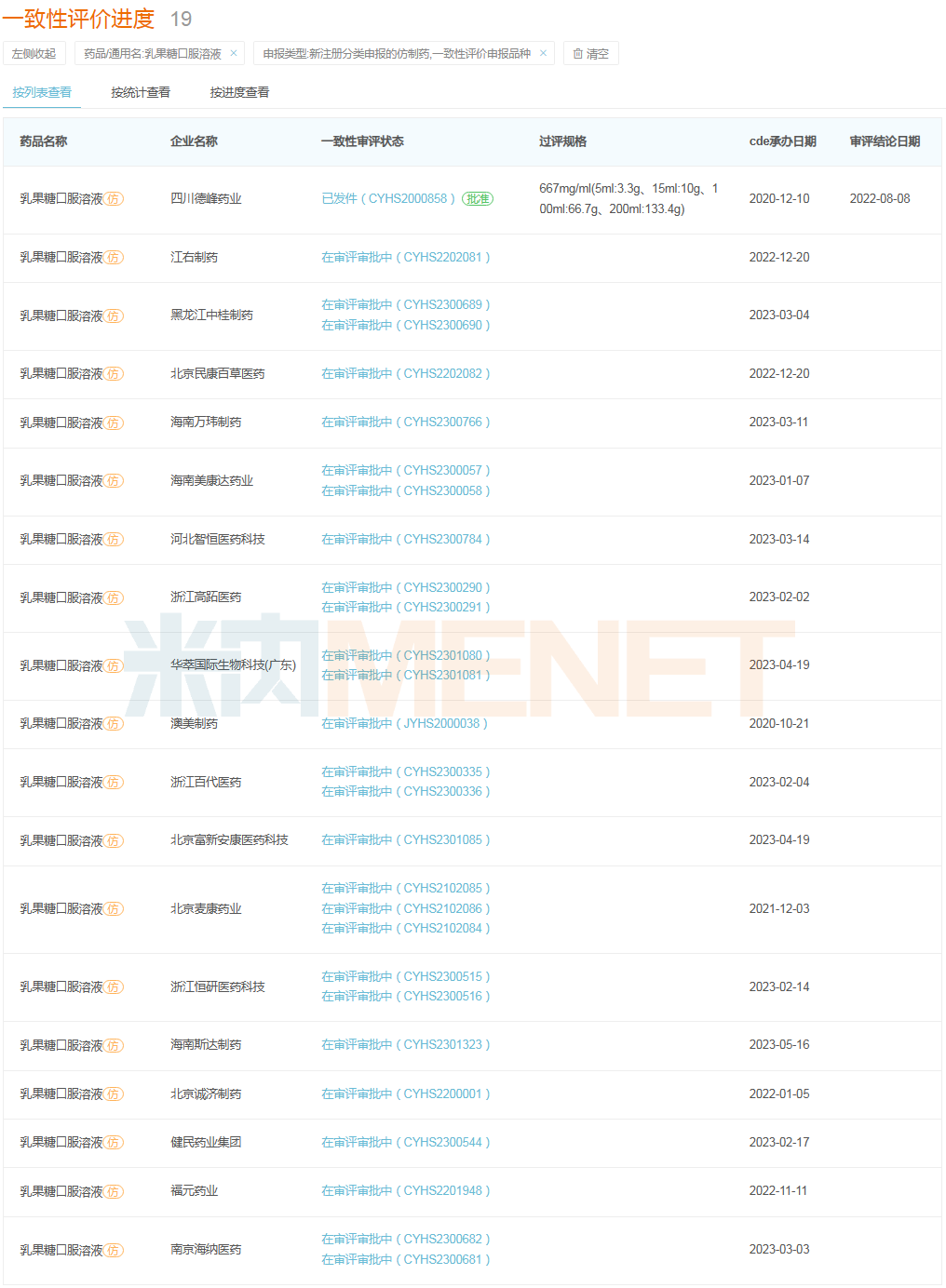
Currently, no company has submitted a supplementary application for consistency evaluation for Lactulose Oral Solution, but 19 companies have submitted marketing applications for the product under the new registration classification. Among them, Sichuan Defeng Pharmaceutical’s product was approved and deemed equivalent to passing the consistency evaluation, making it the first domestic company to do so. It is not difficult to see that some commonly used pediatric prescription drugs, after systematic evaluation, are being switched to OTC, improving the convenience of purchasing medication for children and their families. This also benefits companies with products in this category, allowing them to expand into the retail market.
Status of Consistency Evaluation for Lactulose Oral Solution
Budesonide is a major inhaled drug, and Budesonide Nasal Spray is its second best-selling formulation. In recent years, sales of this drug in China’s retail pharmacy terminal have been expanding annually, exceeding RMB 300 million in 2022. Notably, sales growth in online pharmacies reached 102%, indicating strong market potential. Currently, Budesonide Nasal Spray has manufacturing approvals only from AstraZeneca and Taiwan’s Haimeni Pharmaceutical. Two companies, Nanchang Baiji Pharmaceutical and Sichuan Purent Pharmaceutical, have submitted marketing applications that are under review. For “Rx-to-OTC” products that are exclusive or face less intense competition, the retail pharmacy terminal is poised to become a significant driver of sales growth.
Status of Marketing Applications for Budesonide Nasal Spray Under Review


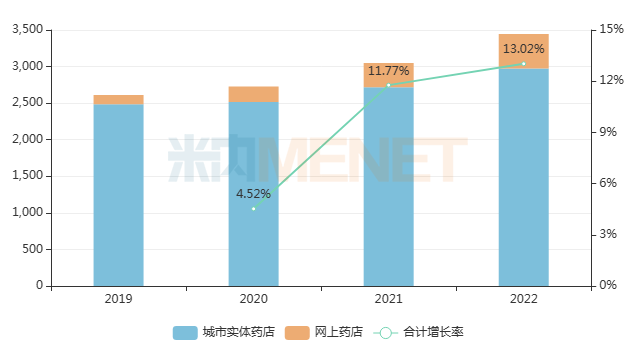
Data shows that the total market size for chemical drugs and TCM patent medicines in China’s retail pharmacy terminal in 2022 was nearly RMB 350 billion, an increase of 13.02% year-on-year. According to incomplete statistics, 813 drugs have been switched from prescription to OTC since 2004, involving thousands of manufacturers. As more prescription drugs are switched to OTC, the retail market is set to face new waves of competition.
Recent Overall Sales Trends of Chemical Drugs and TCM Patent Medicines in China’s Retail Pharmacy Terminal (Unit: RMB 100 million)

Is “Rx-to-OTC” Becoming an Inevitable Trend? What Lies Behind It?
The expansion of “Rx-to-OTC” switches relies on the continuous improvement of China’s drug management system. With the implementation of relevant policies, a situation with multiple benefits is emerging.
1. Benefiting Drug Consumers
According to the classified management requirements for prescription drugs and OTC drugs (Category A/B) under the Drug Administration Law, when a prescription drug is switched to OTC, patients no longer need a doctor’s prescription to purchase it from offline pharmacies or online e-commerce platforms. This increases the channels for purchasing medication and makes it more convenient, particularly benefiting patients requiring long-term medication.
2. Reducing National Medical Insurance Expenditure
Medical and pharmaceutical expenditures impose a financial burden on the government. Drugs switched from prescription to Category B OTC will be removed from the national medical insurance reimbursement system. In the long run, “Rx-to-OTC” switches will be an effective measure to conserve national medical insurance funds.
3. Saving Clinical Diagnosis and Treatment Resources
For some minor ailments, patients can achieve recovery through self-diagnosis and self-medication. This reduces the need for physicians to spend time and effort on prescribing such medications, allowing them to devote more time to treating patients with serious illnesses, improving their own diagnostic and treatment skills, and gaining professional satisfaction and a sense of achievement from their work and patient satisfaction.
4. Expanding Retail Channels and Extending Product Lifecycles
Drugs, like other products, have a lifecycle. When a patented drug expires, it faces fierce price competition from generics, leading to a sharp drop in sales, known as the “patent cliff.” This is compounded by policy impacts such as healthcare cost control and centralized procurement price reductions, which have even led some original drugs to exit the market. Switching a prescription drug to OTC can, on one hand, extend the product’s lifecycle and generate more revenue for the pharmaceutical company. On the other hand, it broadens the target audience from specific patients to the general public, which can help boost sales of other products from the company.
5. Enriching Product Variety and Driving New Growth in the Retail Industry
For retail pharmacies, a wider range of OTC drugs better meets consumer demand. “Rx-to-OTC” drugs, which can be purchased directly in pharmacies, are expected to increase product sales volume and expand market share. If the drug already had a solid consumer base during its prescription stage, switching to OTC opens up new channels, potentially generating even greater profits for the company. In recent years, China’s OTC market has grown rapidly. “Rx-to-OTC” varieties gain access to more flexible promotion and sales channels, presenting significant development opportunities for the OTC market and related companies.
Conclusion
It can be foreseen that factors such as centralized procurement, healthcare cost control, and pharmaceutical reforms will drive a significant number of prescription drugs out of hospitals. Industry insiders speculate that in the future, about 70% of conventional prescription drugs will be converted to non-prescription drugs. These drugs will gradually move to the off-hospital market, unlocking significant market potential. “Riding on the policy tailwind of prescription-to-OTC switches, how to quickly leverage the advantages of prescription drugs into the OTC market, providing consumers with products that are more professional, with clearer efficacy, and a better user experience” will become a key focus of competition for companies in the next phase.