Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2022-03-15
Posting Date:2022-03-15 Views:
Views: 01 The Sixth Round of National Centralized Procurement to Be Implemented
On March 8, Quanyaowang (Shenzhen Drug Trading Online Platform) issued the "Notice on Carrying out the Declaration of Selected Drugs for the Sixth Round of National Organized Drug Centralized Procurement (Insulin Special)."

According to the notice, the registration deadline for the procurement platform is 5:30 PM on March 31, 2022. Based on the pattern of previous centralized procurement, after the completion of preparatory work such as listing, the selected results will be formally implemented shortly.
Since the announcement of the selected list for the sixth round of national centralized procurement in November 2021, multiple provinces have been preparing for its implementation. According to preliminary statistics from Sinaleo, Shanghai, Heilongjiang, and Gansu have all issued notices related to listing.
According to previous information from the National Healthcare Security Administration, the sixth round of national centralized procurement will be formally implemented in May. The early listing by multiple provinces and cities is expected to prepare for the subsequent implementation and procurement of the selected products. Based on past experience, on the official implementation date, the selected products will generally become available for procurement from 12:00 AM. This indicates that the implementation of this special insulin round of national centralized procurement is imminent.
Shanghai
On February 28, the Shanghai Sunshine Pharmaceutical Procurement Network issued the "Notice on Carrying out the Listing of Selected Drugs from the National Drug Centralized Procurement (Insulin Special)." It stipulated that from March 1, 2022, enterprises log in to the "Shanghai Pharmaceutical Procurement Service and Supervision Information System," enter the "Drug Declaration" sub-column, select "National Centralized Procurement Selected Varieties" under the listing type, and report information on selected drugs for the Shanghai region. The deadline for declaration is March 31.
Heilongjiang
On February 17, the Heilongjiang Provincial Drug Centralized Procurement Center issued a notice on carrying out the listing of selected drugs from the sixth round of national organized drug centralized procurement (insulin special).
According to the requirements of the Heilongjiang Provincial Procurement Center, the submission period for relevant drug manufacturers is from 9:00 AM on February 18 to February 28. The price to be reported should be the price per smallest unit of the selected drug.
Gansu
On December 21, 2021, the Gansu Provincial Public Resources Trading Center issued the "Notice on Publishing the Results of the Listing Declaration for Selected Drugs from the Sixth Round of National Organized Drug Centralized Procurement (Insulin Special)." It stated that after enterprise declaration and review by the provincial drug procurement agency, a total of 78 specifications for selected drugs from the sixth round of national centralized procurement (insulin special) were declared for listing.
Following the announcement of the review results, formal listing of the procurement results is expected to occur shortly.
It is now March, leaving only two months until the implementation of the sixth round of centralized procurement.
02 Market Substitution Set to Begin
According to CCTV News, 32,000 medical institutions participated in this round of national procurement, declaring a demand for approximately 200 million units of insulin, involving a value of nearly 17 billion yuan.
Based on the results of the sixth round of national procurement, the actual agreement procurement volume is approximately 14 billion yuan. According to Mnet data, sales of insulin and its analogues in China's public medical institution terminals were close to 27 billion yuan in 2020. Overall, outside of this procurement round, there remains a market competition worth over 10 billion yuan.
In this round of national procurement, domestic pharmaceutical companies emerged as the biggest winners. For example, Tonghua Dongbao holds the largest share of the second-generation insulin market, with a total demand of 31.39 million units, accounting for 34.78% of the total second-generation insulin demand. Gan & Lee Pharmaceuticals holds the largest share of the third-generation insulin market, with a total demand of 11.60 million units, accounting for 9.39% of the total third-generation insulin demand.
Foreign companies have long dominated China's insulin market competition. Novo Nordisk, Sanofi, and Eli Lilly collectively hold nearly 75% of the market share.
When the results of the sixth round of national centralized procurement (insulin special) were announced, many industry experts noted that the launch of insulin centralized procurement could promote the substitution of domestic brands, potentially further expanding the market share of domestic enterprises.
Judging from the current progress of the sixth round of national centralized procurement (insulin special), domestic pharmaceutical companies are indeed striving hard, gearing up for the procurement. After six of its products won bids in this round, Gan & Lee Pharmaceuticals announced plans to double its marketing team, expanding from its current size of one thousand. Meanwhile, Gan & Lee also stated on its investor interaction platform that after the centralized procurement, the price of insulin in the out-of-hospital market would be adjusted accordingly with reference to the procurement price.
At the same time, multinational pharmaceutical companies are not to be outdone. For instance, Eli Lilly is constructing a new production line at its insulin plant, and its insulin product group continues to recruit pharmaceutical representatives.
On March 5, the State Council Premier clarified in the government work report the need to advance the centralized volume-based procurement of drugs and high-value medical consumables, ensuring production and supply.
Having come this far, as the first biological drug to enter national centralized procurement, the insulin procurement will also face practical tests involving medical institutions, patients, and other stakeholders. Whether domestic insulin can successfully achieve import substitution and how the insulin market will further differentiate remain open questions.
The sixth round of national centralized procurement (insulin special) opens a window for the procurement of biological drugs. Currently, an increasing number of biological drugs are facing patent expirations, and the number of marketed biosimilars is also increasing. Statistics show that there are nearly 500 biosimilars under development in China, with nearly 50 expected to be launched by this year.
Due to factors such as complex production and application technologies, the industry has generally believed that differences exist in efficacy and safety between biosimilars and original drugs. It is well known that the prices of biological drugs have generally been relatively high.
Chen Jinfu, Deputy Director of the National Healthcare Security Administration, revealed at a regular State Council briefing that while the quality evaluation methods for biosimilars differ from those for generic chemical drugs, they adhere to strict quality standards, and their inclusion in centralized procurement is undoubtedly the next step.
Considering multiple factors, the centralized procurement of biological drugs has become quite necessary.
There are no forbidden zones for centralized procurement, and biological drug enterprises should prepare early. Currently, ensuring production capacity appears to be the most critical aspect for biosimilar procurement.
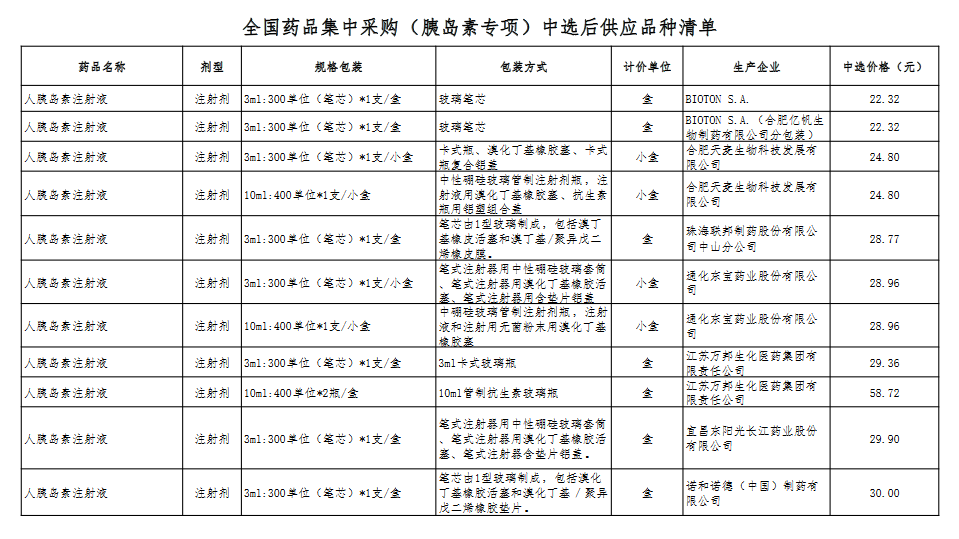
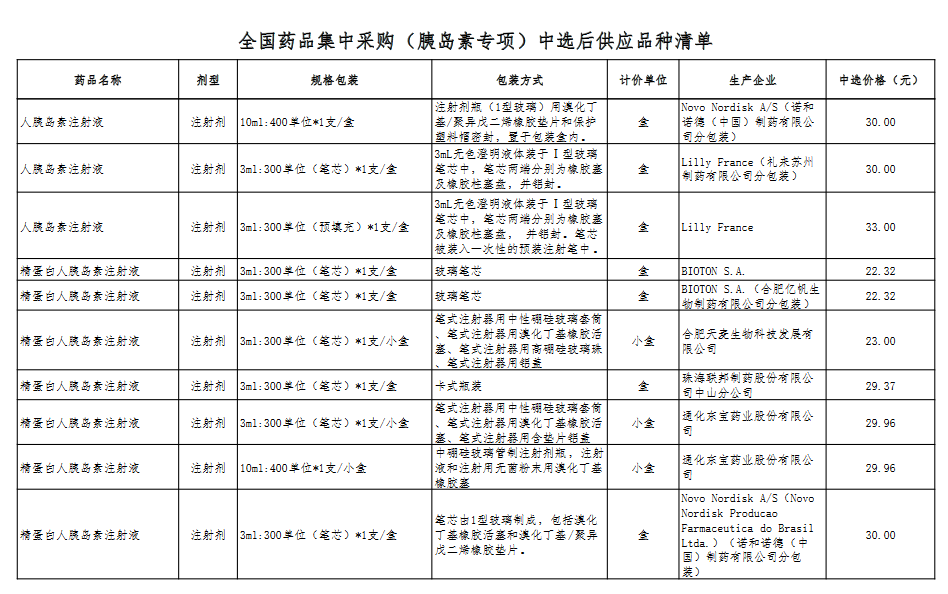
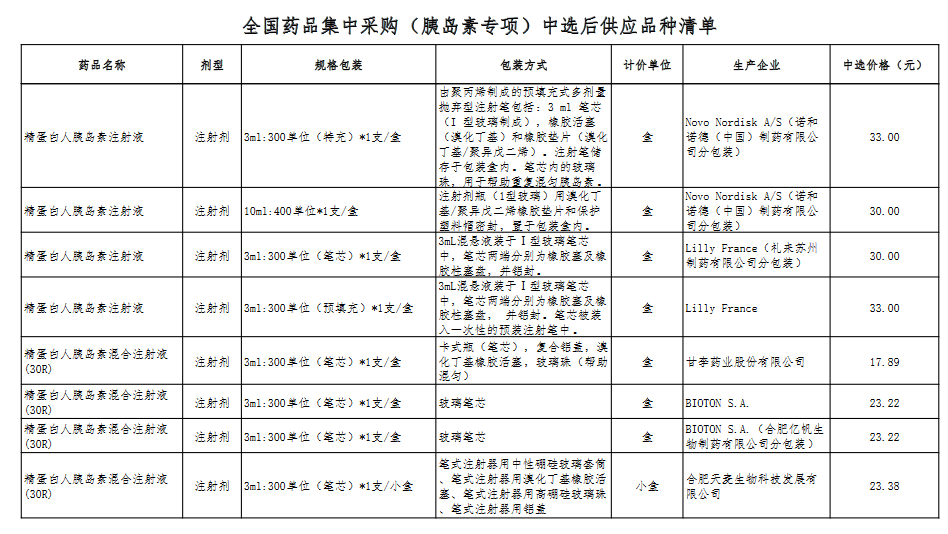
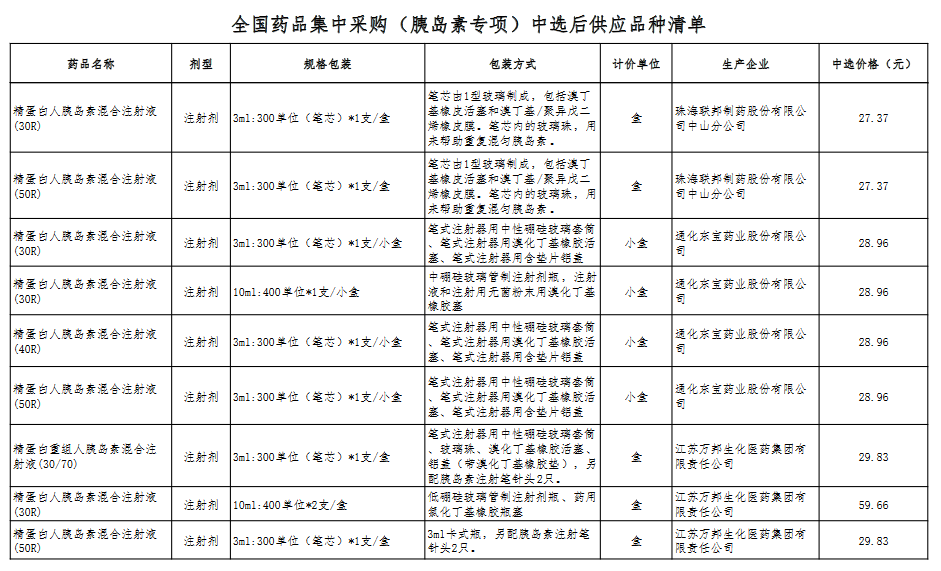
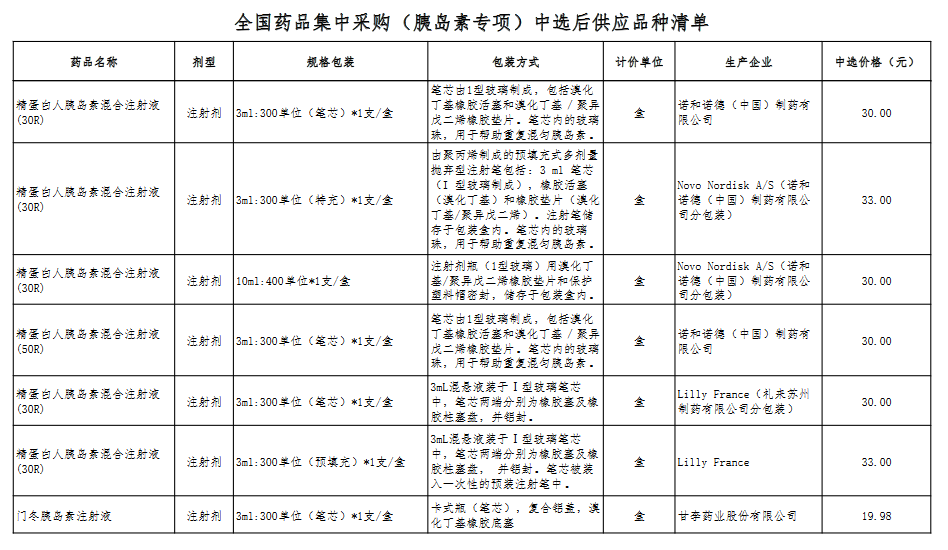
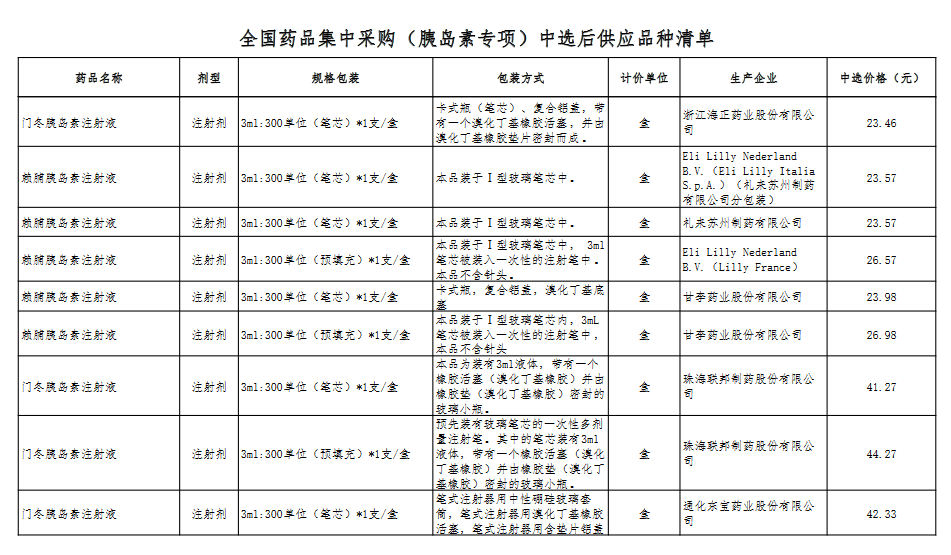
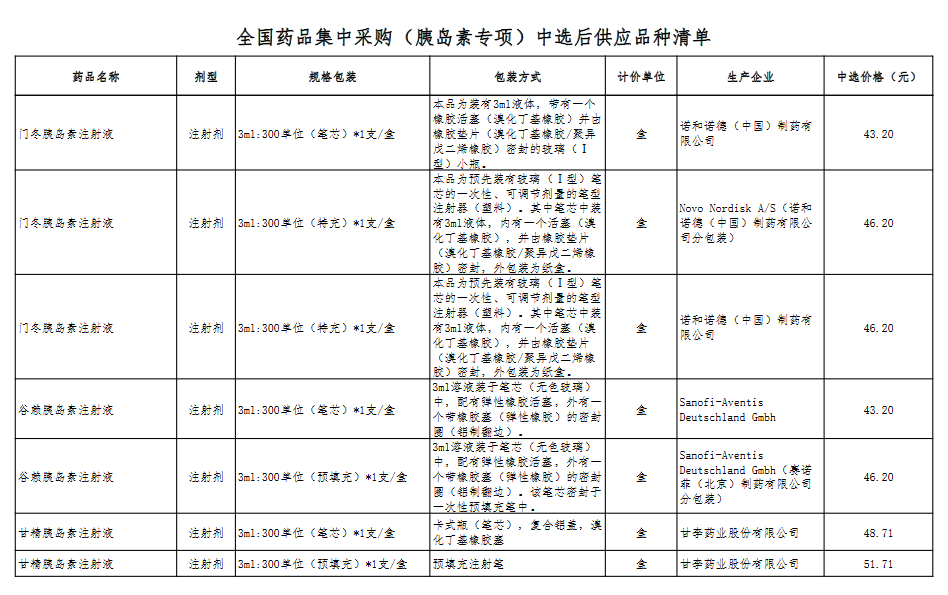
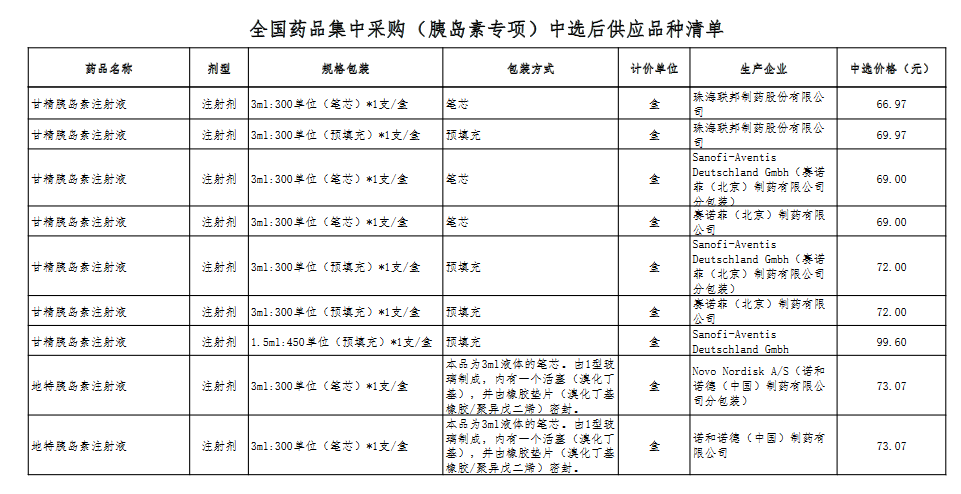
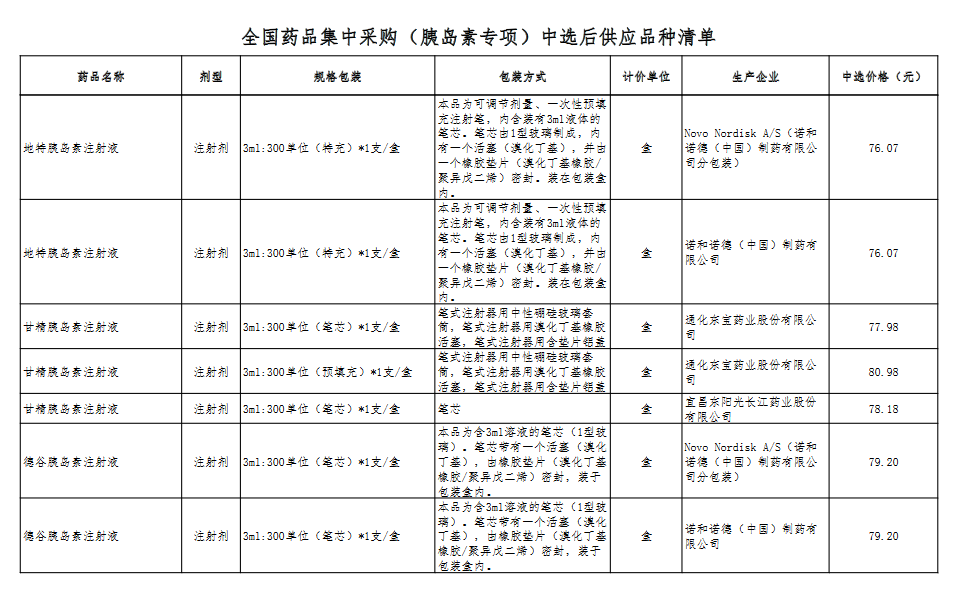
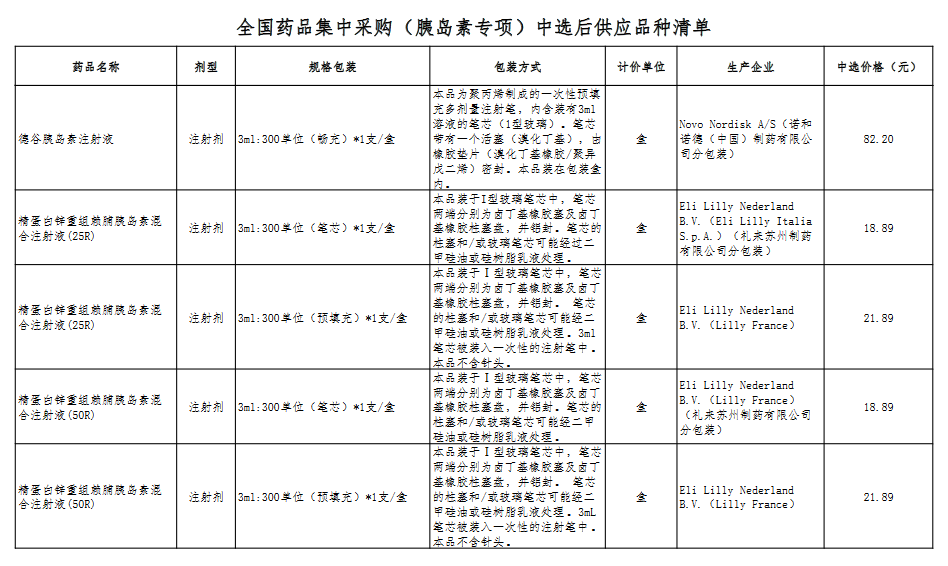
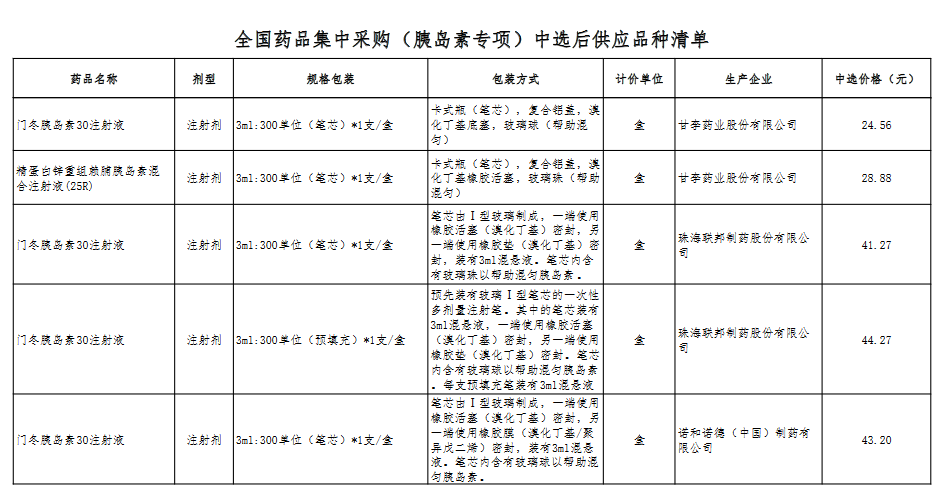
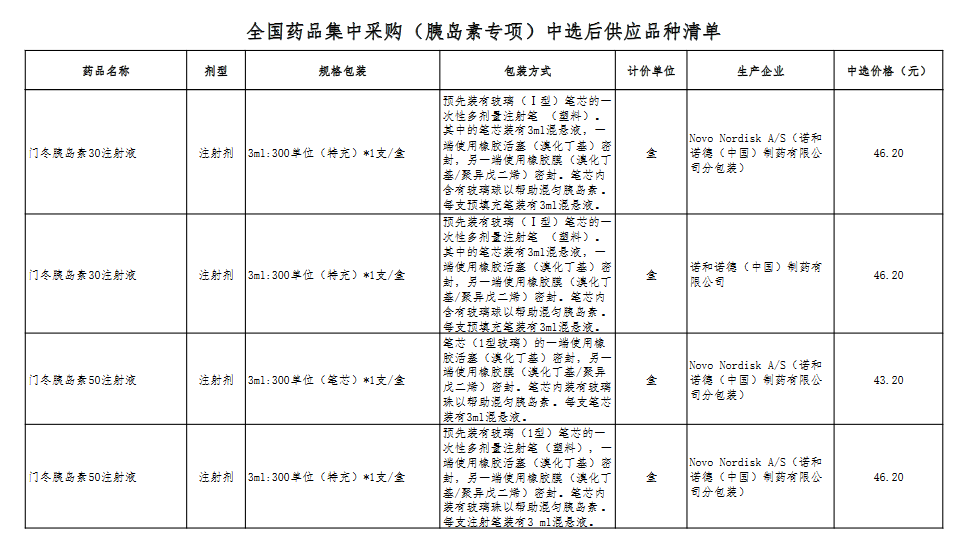
Attachment: Quanyaowang's National Drug Centralized Procurement (Insulin Special) List of Supply Varieties After Bid Selection