



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2022-01-28
Posting Date:2022-01-28 Views:
Views: Recently, Sierra Oncology announced positive topline results from the pivotal Phase 3 MOMENTUM study. This global, randomized, double-blind clinical trial evaluated the efficacy and safety of momelotinib (MM) compared with danazol (DAN) in patients with symptomatic and anemic myelofibrosis (MF) who had been previously treated with an approved JAK inhibitor. Results showed that the study met all primary and key secondary endpoints.
Momelotinib is a potent, selective, orally bioavailable inhibitor of JAK1, JAK2, and ACVR1/ALK2. In 2018, Sierra Oncology acquired the drug from Gilead Sciences for $198 million (including milestone payments).
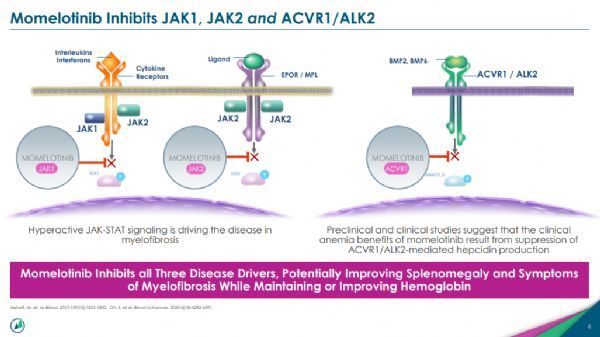
Mechanism of Action of Momelotinib
Momelotinib is currently being developed for the treatment of patients with symptomatic and anemic myelofibrosis who have been previously treated with an approved JAK inhibitor. Since the initiation of clinical studies in 2009, over 1,200 patients have been treated with momelotinib, including approximately 1,000 patients treated for myelofibrosis, with some patients receiving treatment for more than 11 years.
Notably, momelotinib is the first and only JAK inhibitor to demonstrate positive data across all key markers of disease symptoms, spleen response, and anemia.
Topline data from 195 patients (BBM n=130; DAN n=65) include:
——Total Symptom Score (TSS) reduction >50% (primary endpoint): 25% of patients in the MMB treatment group achieved this goal compared to 9% in the control group (p=0.0095);
——Transfusion independence (TI, secondary endpoint): 31% of patients in the MMB treatment group achieved this goal compared to 20% in the control group (one-sided p=0.0064; non-inferiority);
——Spleen Response Rate (SRR) >35% (secondary endpoint): 23% of patients in the MMB treatment group achieved this goal compared to 3% in the control group (p=0.0006);
——During the randomized treatment period, the incidence of grade 3 or higher severe adverse events was 54% in the MMB treatment group and 65% in the control group. Serious treatment-emergent adverse events occurred in 35% and 40% of patients in the MMB treatment and control groups, respectively.
——Mean baseline characteristics across all patients were TSS of 27, hemoglobin level (Hgb) of 8 g/dL, and platelet count of 145 x 10E9/L.
```