



Copyright © 2025 Shenyang Dasan Pharmaceutical Technology Co., Ltd.





 Posting Date:2023-03-17
Posting Date:2023-03-17 Views:
Views: Here is the translation of the website content into English:
Recently, frequent news has emerged regarding national centralized drug procurement (VBP): regions such as Yunnan, Henan, and the Yangtze River Delta (Shanghai, Zhejiang, Anhui) have successively initiated contract renewal work for drugs whose national VBP agreements are expiring. This involves over 100 selected varieties, including star products like Abiraterone, Simvastatin, Paclitaxel, and Ambroxol. The bidding for the eighth round of national VBP is approaching its final stage. Data from Menet shows that the 40 shortlisted varieties had total sales exceeding 60 billion yuan in China's public medical institutions in 2021. Among them, 12 varieties are highly competitive, with 10 or more qualified enterprises vying for each. Key players include Beite Pharmaceutical, Fosun Pharma, and Yangtze River Pharmaceutical Group.
VBP Introduces "One Primary, One Backup" Supply System, Procurement Cycle Extended
In November 2022, the Shanghai Sunshine Pharmaceutical Procurement Network released documents for a new round of Yangtze River Delta (Shanghai, Zhejiang, Anhui) Alliance Centralized Procurement (hereinafter referred to as the "Alliance Procurement"). After a tumultuous process lasting nearly five months, the final selection results were officially announced in March 2023.
According to the procurement requirements, this Alliance Procurement covers Shanghai, Zhejiang, and Anhui. The selected products consist of 43 varieties whose national VBP procurement periods were nearing expiration (originally 44 were included; Cefradine oral solid dosage form was apparently not awarded). The agreed procurement volume for the first year for each variety is set at 60% of the baseline procurement volume for each region.
The procurement period for selected products is similar to that of the eighth round of national VBP, with the renewal procurement period extended until the end of 2025, approximately a three-year term. This suggests that the end of 2025 marks not only the conclusion of the 14th Five-Year Plan but also the target period for各地 to complete their VBP implementation goals.
Furthermore, the procurement rules are familiar, employing a comprehensive scoring system that evaluates both drug-specific and enterprise-specific dimensions. The highest-scoring bidder is selected, indicating that a low price is not the sole advantage. Simultaneously, the "One Primary, One Backup" mechanism from the seventh round of national VBP was introduced. This means that within a single alliance region, for a specific variety, one enterprise is designated as the primary supplier. Additionally, a second-ranked enterprise that did not originally supply the region under the national VBP but achieved a high comprehensive score is designated as a backup supplier. Should the backup enterprise decline to supply, the backup role is filled by the next highest-ranked enterprise based on the comprehensive score.
Detailed Comprehensive Scoring Criteria
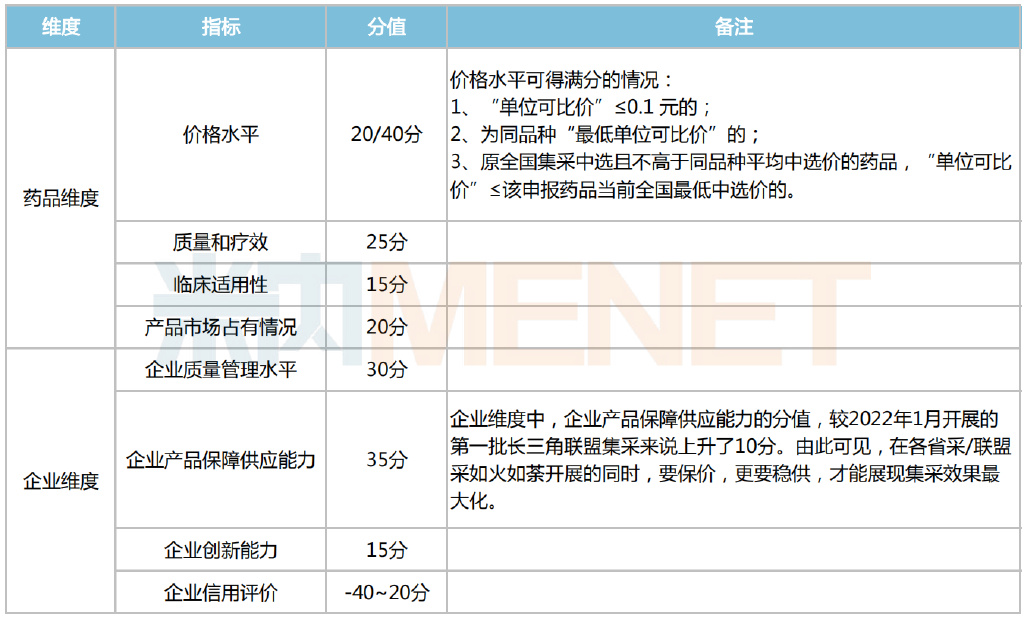
It is noteworthy that the backup supplier can list their product on the procurement platform at their quoted price. If the backup supplier was a previous national VBP winning supplier for that product, they can list at the lower of their current quoted price or their previous national VBP winning price. If the primary supplier cannot meet the market demand in the supply region, the backup supplier can assume the role of the primary supplier through the relevant procedures. This mechanism significantly reduces the probability of supply disruptions for the selected drugs, creating a "double guarantee" for supply.
Billion-Yuan Market Reshuffled! Yangtze River, Hengrui, and Others Achieve Significant Gains
A total of 43 drugs were selected in this Alliance Procurement, all of which were previously included in national VBP rounds, specifically the second and fourth rounds. By therapeutic category, the selected varieties are primarily cardiovascular system drugs, antineoplastic and immunomodulating agents, and digestive system and metabolism drugs, each accounting for six or more selections.
Selected Varieties in the Shanghai-Zhejiang-Anhui Alliance Centralized Procurement
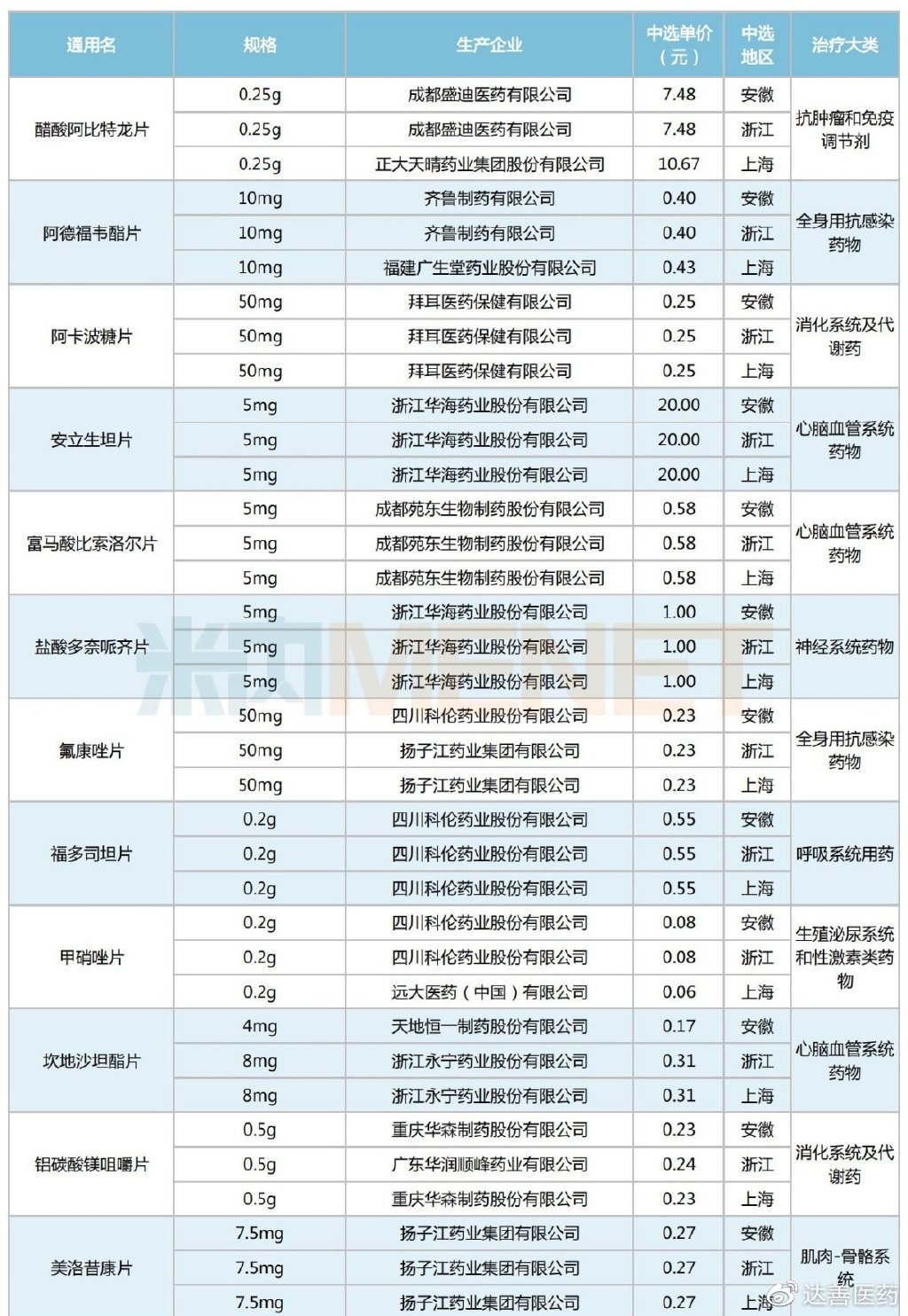
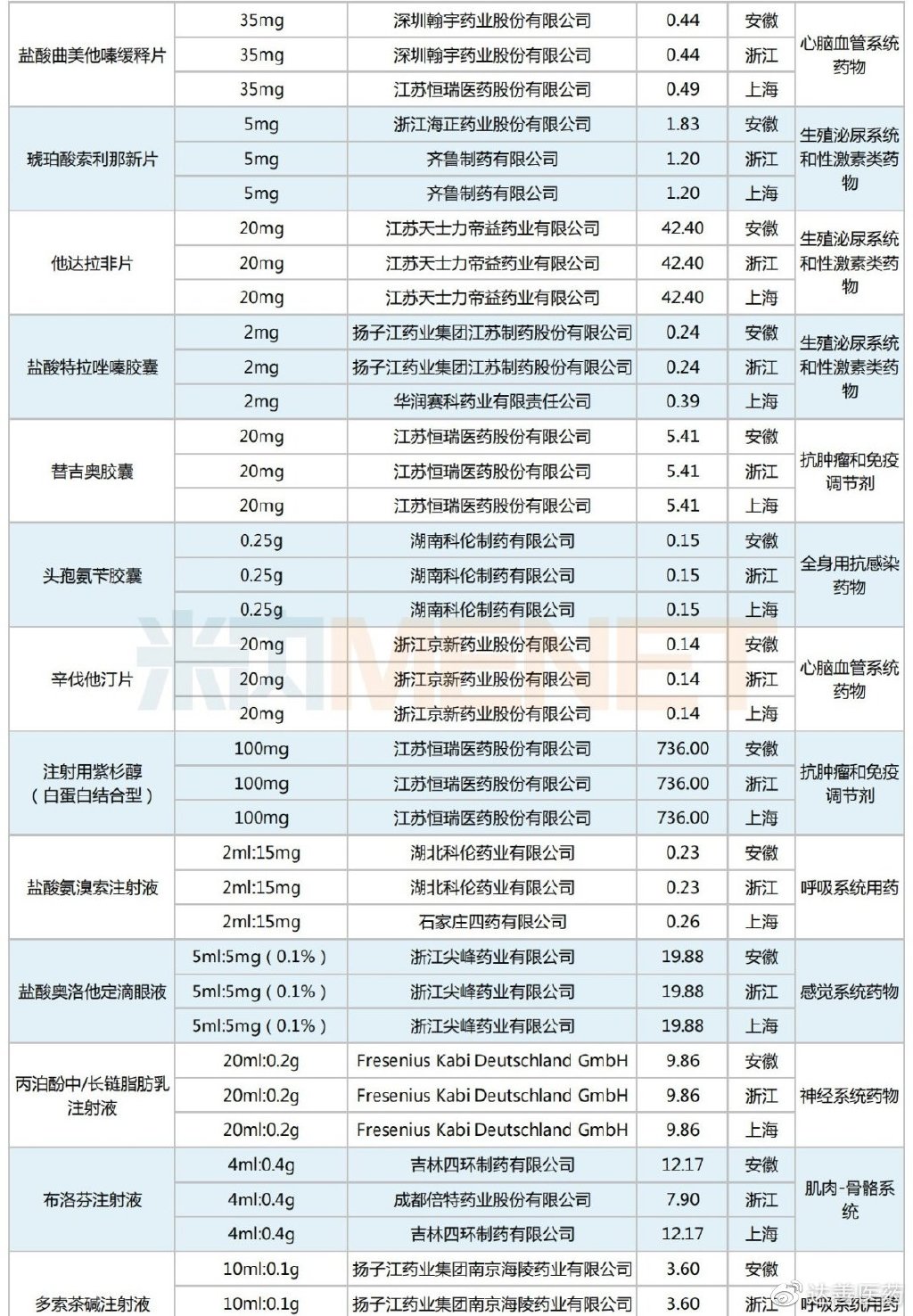
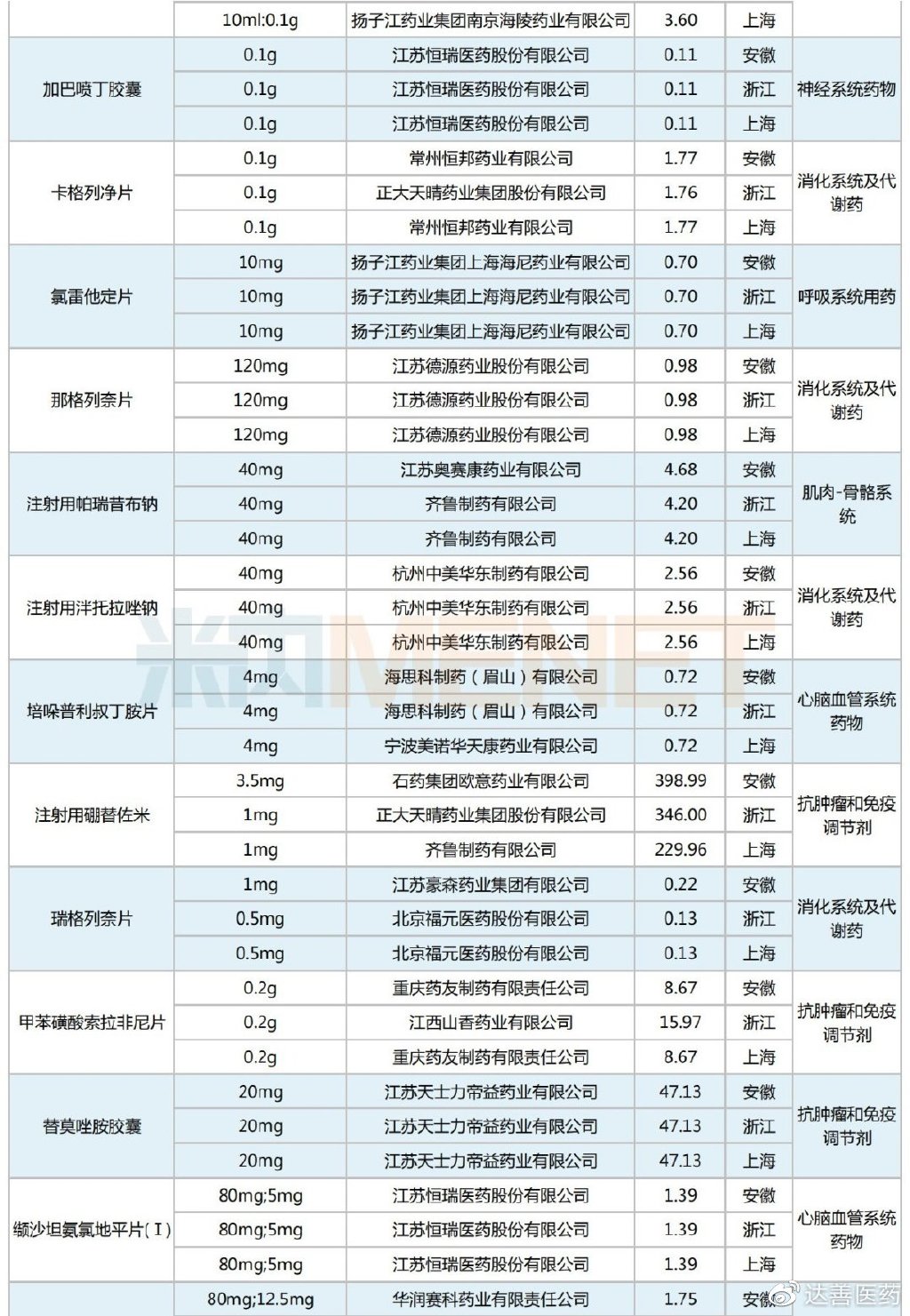
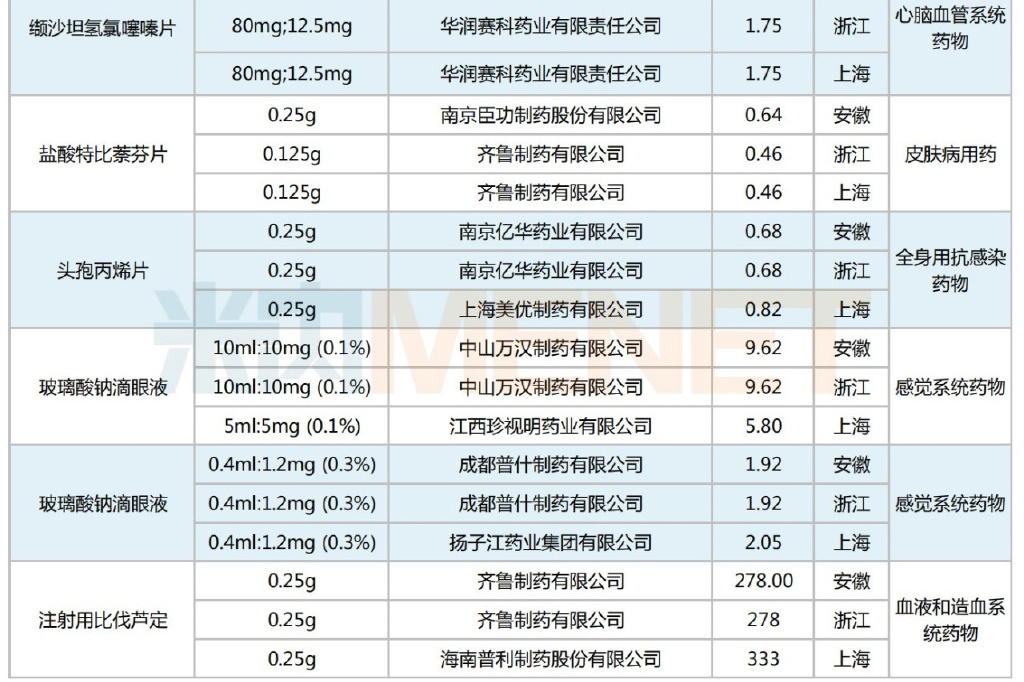
Regarding selected prices, compared to the original national VBP winning prices, this Alliance Procurement still saw predominantly price reductions. However, there were 17 products that experienced price increases, including Yuan Dong Pharmaceutical's Bisoprolol Fumarate Tablets, Jingxin Pharmaceutical's Simvastatin Tablets, Qilu Pharmaceutical's Parecoxib Sodium for Injection, Kelun Pharmaceutical's Metronidazole Tablets, and Bayer Pharma's Acarbose Tablets. The average price increase was 51%, with the highest single product increase reaching 133%.
Jingxin Pharmaceutical's Simvastatin Tablets performed notably well, successfully winning selection with a 133% price increase compared to its national VBP winning price. This reflects that winning a national VBP bid does not solely rely on price cuts; adjustments can be made based on comprehensive factors such as product efficacy and clinical demand, aiming to maximize benefits for both enterprises and patients.
Selected Products with Price Increases in the Alliance Procurement
In terms of winning enterprises, Yangtze River Pharmaceutical Group, Hengrui Medicine, Qilu Pharmaceutical, and others each had 10 or more products selected (counting products across different winning provinces).
Yangtze River Pharmaceutical Group had 14 products selected in this Alliance Procurement, including Fluconazole Tablets, Meloxicam Tablets, Terazosin Hydrochloride Capsules, Doxofylline Injection, Loratadine Tablets, and Sodium Hyaluronate Eye Drops. Notably, Yangtze River's Doxofylline Injection held approximately 31% of the public hospital market in Shanghai, Zhejiang, and Anhui during Q1-Q3 2022, ranking second among brands. With Yangtze River exclusively securing the supply rights for these three provinces, it is poised to further accelerate its market expansion.
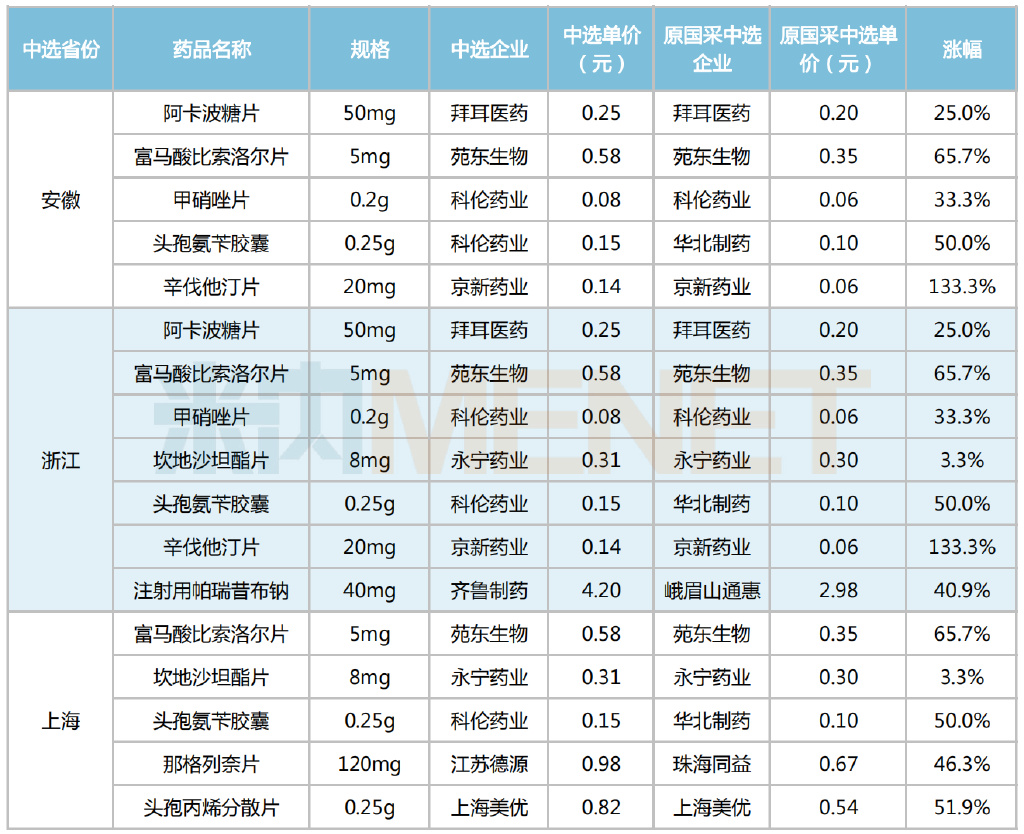
Hengrui Medicine had 13 products selected, including Trimetazidine Hydrochloride Sustained-Release Tablets, Tegafur Gimeracil Oteracil Potassium Capsules, Paclitaxel for Injection (Albumin-Bound), Gabapentin Capsules, and Valsartan Amlodipine Tablets (I). Among these, Paclitaxel for Injection (Albumin-Bound) achieved sales exceeding 180 million yuan in public hospitals across Shanghai, Zhejiang, and Anhui in 2021, a year-on-year increase of 28.15%. Hengrui Medicine held over 70% of the market share, solidifying its position as the leading brand.
2021 Brand Landscape for Paclitaxel for Injection (Albumin-Bound) in Public Hospitals in Shanghai, Zhejiang, and Anhui
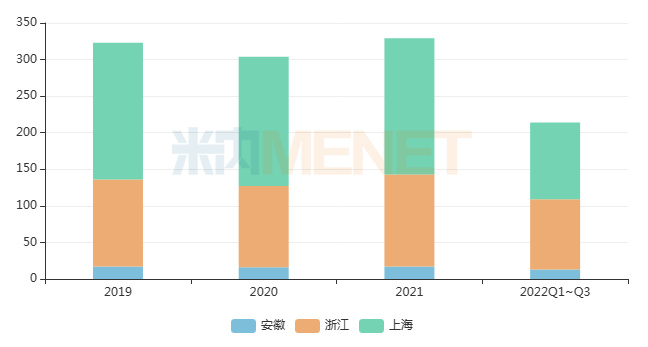
Data from Menet indicates that in the key provincial public hospital market, total sales in Shanghai, Zhejiang, and Anhui exceeded 30 billion yuan annually from 2019 to 2021. This round of Alliance Procurement included 43 drugs, with total sales in these public hospital terminals exceeding 1 billion yuan in 2021.
Recent Sales Scale in Public Hospitals in Shanghai, Zhejiang, and Anhui (Unit: 100 million yuan)
Eighth Round of National VBP Bidding Imminent, Beite, Fosun, and Others Poised for Action
On one hand, provincial contract renewal work for national VBP is progressing vigorously; on the other hand, the bid opening date for the eighth round of national VBP is drawing near.
In early March, the Shanghai Sunshine Pharmaceutical Procurement Network officially released the document for the eighth round of national centralized drug procurement. The new round of bids is scheduled to open on March 29th in Lingshui County, Hainan Province. It encompasses 40 varieties (106 specifications). Menet data shows that the total sales of these 40 varieties in China's public hospitals, county-level public hospitals, urban community centers, and township health centers (collectively referred to as China's public medical institutions) exceeded 60 billion yuan in 2021. Among these, 12 varieties are highly competitive, with 10 or more enterprises (counted by group; multiple subsidiaries of the same group that have passed generic consistency evaluation are counted as one) vying for selection.
Among the 40 varieties, there are 10 domestic enterprises (by group) that have 5 or more evaluated varieties. These include well-known major pharmaceutical companies such as Beite Pharmaceutical, Fosun Pharma, Yangtze River Pharmaceutical Group, Kelun Pharma, China Resources Pharma, Qilu Pharmaceutical, China Biopharmaceutical, and Shandong New Times.
Looking at dosage form distribution and therapeutic areas, 27 of the 40 varieties are injections, accounting for over 65%. Ten are oral solid dosage forms, and there is one each for granules, sustained-release controlled-release forms, and oral solutions. They span 8 therapeutic categories, primarily systemic anti-infectives, cardiovascular system drugs, and blood and hematopoietic system drugs, each with 6 or more varieties included in the eighth round of national VBP.
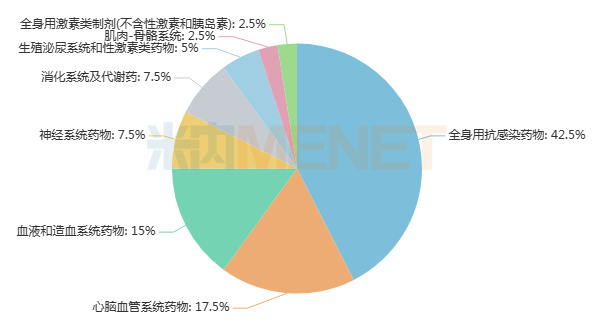
Distribution of the 40 Varieties by Therapeutic Category
Furthermore, the eighth round of national VBP documents also stipulate: ① The procurement period for each variety runs from the execution date of the winning results until December 31, 2025. Under this unified procurement period, for varieties with fewer winning enterprises, it effectively extends the procurement cycle; ② A "second backup" supply mechanism is added for some varieties prone to clinical supply shortages, creating "multiple layers of insurance" for supply; ③ Given the large number of injection varieties in the eighth round, rules for calculating the winning prices for injections have been re-standardized; ④ Strict measures are to be implemented to crack down on violations such as "bid-rigging, collusive bidding, and fraud" during the procurement process...
Overall, the eighth round of national VBP builds upon the experience of previous rounds, continuously optimizing product selection, procurement cycles, and supply backup arrangements, making the VBP rules more scientific and humane.